Commentary: A Stepwise Approach to the Treatment of Lichen Planopilaris
John Plante*, Chelsea Eason, Alan Snyder, Dirk Elston
Department of Dermatology and Dermatologic Surgery, Medical University of South Carolina, Charleston, South Carolina
Lichen planopilaris (LPP) is a primary cicatricial alopecia characterized by pruritus, tenderness, follicular spines and erythema in the active phase1-3. Although the precise etiology is unknown, the pathogenesis involves chronic, lymphocyte-mediated inflammation that may ultimately lead to irreversible, scarring hair loss through its selective destruction of the hair follicle1,2. Three variants of LPP exist and include the classic form, frontal fibrosing alopecia (FFA), and Graham-Little-Piccardi-Lassueur syndrome (progressive alopecia of the scalp, axillae, brows, and pubis)2. LPP often proves refractory to standard therapies and follows an unpredictable, relapsing course, frequently leading to a severely diminished quality of life1,2,4,5. Here we further discuss our recent study concerning the use of tofacitinib in the treatment of recalcitrant LPP6.
Numerous therapies have been used to treat LPP with variable success1-3,5,7-9. Since currently available treatments do not typically lead to hair regrowth, management is directed toward decreasing the rate of hair loss, controlling symptoms, and reducing inflammation2,9. First-line therapies include intralesional and high-potency topical corticosteroids, but these can result in scalp atrophy and compromise the success of future hair transplantation. Other topical agents have less risk of atrophy and include the calcineurin inhibitors tacrolimus and pimecrolimus2. Although it does not control the underlying inflammatory process, both topical and oral minoxidil can also be used to maximize hair regrowth in patients who also suffer from inherited pattern alopecia – almost 50% of the population9,10. Additional systemic therapies include hydroxychloroquine, mycophenolate mofetil, cyclosporine, methotrexate, thalidomide, doxycycline, retinoids, pioglitazone, 5-alpha reductase inhibitors, and short-term prednisolone1,2,9. Recent studies have suggested the potential of low-dose oral naltrexone, platelet-rich plasma injections, and non-pharmacologic interventions such as excimer laser, but none have been uniformly effective5,7,8. While hair pieces and extensions may increase volume and cover alopecic areas, caution must be exercised to avoid traction9.
In a recently published study, we investigated the potential of topical and oral (systemic) formulations of tofacitinib6. Tofacitinib is a Janus kinase (JAK) 1/3 inhibitor with efficacy against several immune-mediated dermatologic conditions, including atopic dermatitis, alopecia areata, and vitiligo11-14. Its immunomodulating activity is mediated through inhibition of the JAK/STAT signaling pathway12,13. In LPP, interferons have been implicated in facilitating lymphocytic recruitment and the loss of bulge epithelial cell immune privilege1. Through tofacitinib’s effects on the JAK/STAT pathway, this interferon-mediated inflammation may be reduced, thereby improving symptoms and preventing further follicular destruction1,11-13.
We explored the therapeutic potential of tofacitinib in the treatment of refractory LPP through a retrospective chart review6. To the authors’ knowledge, this is the second study investigating tofacitinib’s use in LPP management1. Our study included nine patients who had failed other therapy – three used topical, five used oral, and one used both formulations.
A favorable initial response was achieved in all but one patient. The remaining subject responded poorly to topical therapy but improved with systemic tofacitinib therapy. Although the absolute number of patients is small, there was a trend toward better clinical response in those on systemic as compared to those on topical therapy. The time to a favorable response ranged from 1 to 7 months, with 5 and 8 patients demonstrating improvement within the first 3 and 5 months of treatment, respectively.
Our current practice pattern is to treat rapidly progressive disease with a short course of prednisone or cyclosporine and then try to transition to topical calcineurin inhibitor therapy when possible (Figure 1). A subset of patients will achieve remission of their disease with pioglitazone at doses of 15-45 mg daily. Patients should be advised of the labeled indication, mixed data regarding efficacy, and boxed warning about bladder cancer with prolonged use. Our practice is not to continue pioglitazone after a maximum of 9 months with increasing doses in increments. Adjunctive agents include naltrexone at a dose of 5 mg daily and N-acetylcysteine at doses of 600-1200 mg three times daily. Excimer laser, hand-held narrow-band UVB, or low intensity laser cap therapy is often used as adjunctive therapy in those with a partial response. Hydroxychloroquine can occasionally be of benefit, but most of the patients referred to our clinic have already failed hydroxychloroquine therapy. Those who fail to respond may require treatment with methotrexate, mycophenolate mofetil, or tofacitinib. The results of our recent study lend additional support to the idea that both topical and systemic tofacitinib may be viable adjunctive treatment modalities in refractory cases. Hair disorders often respond slowly, and we re-evaluate at 3-month intervals to determine response to therapy and need for escalation.
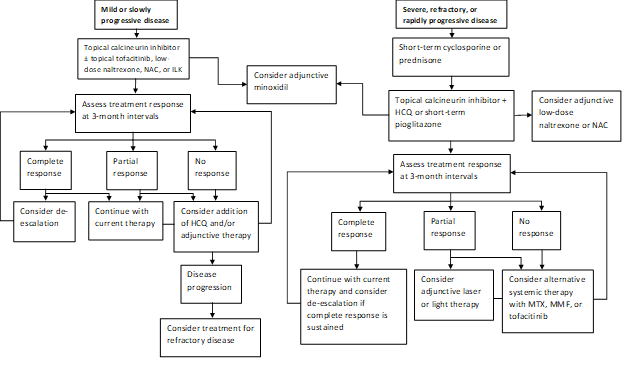
Figure 1: Stepwise Treatment Algorithm for LPP
NAC, N-acetylcysteine; ILK, intralesional corticosteroids; HCQ, hydroxychloroquine; MTX, methotrexate, MMF, mycophenolate mofetil
When considering tofacitinib’s use in practice, practitioners must balance the benefits of this medication with its risks. Our cohort only experienced minor laboratory abnormalities (one on topical and two on systemic therapy) that did not require intervention. However, providers should be aware of the potential side effects when prescribing this medication, including an increased risk of infection, cytopenias, hyperlipidemia, liver and renal abnormalities, as well as the potential for thromboembolic events14-16. While the topical formulation has demonstrated lower plasma concentrations than its systemic counterpart, minor adverse events and laboratory abnormalities have still been reported with topical therapy13.
While our results are promising, they must be interpreted after considering several limitations. We used a retrospective study design, and were not able to employ a validated measure of disease severity such as the Lichen Planopilaris Activity Index (LPPAI)1,3. The LPPAI is suitable for prospective studies and provides a standardized tool by which providers can directly compare outcomes across various treatment modalities. Signs and symptoms such as erythema, tenderness, presence of follicular spines, and affected area were used to assess the therapeutic response, but future studies should also include a measure of quality of life. The possibility of observer bias cannot be excluded as our study was unblinded and non-randomized.
In addition, our entire cohort used tofacitinib adjunctively, making it difficult to assess the independent contribution of the drug. In a prior study, a portion (N=5) of the cohort used tofacitinib as monotherapy, and most subjects achieved favorable outcomes, but this study was also limited by its small sample size and retrospective design1.
Current studies are limited by their small sample sizes and retrospective study designs, and well-designed prospective studies are badly needed. We advise a stepwise approach to therapy to minimize the risk of adverse effects. The risk of permanent scarring must be weighed against the risk of serious adverse events in light of individual patient factors and comorbidities, and patients should be active partners in decision-making. Hair transplantation may be considered once disease control has been achieved and inflammation subsided. It is important to note that aggressive therapy such as prednisone or cyclosporine is typically needed during the weeks following transplantation to avoid flares of disease. Gradual attrition of transplanted hairs is to be expected, even after successful transplantation, but data suggest patients are generally satisfied with the results and find that transplantation improves quality of life.
Abbreviations
LPP, lichen planopilaris; FFA, frontal fibrosing alopecia; JAK, Janus kinase; STAT, signal transducer and activator of transcription proteins; LPPAI, Lichen Planopilaris Activity Index.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Yang CC, Khanna T, Sallee B, et al. Tofacitinib for the treatment of lichen planopilaris: A case series. Dermatol Ther. 2018; 31(6): e12656. doi:10.1111/dth.12656
- Babahosseini H, Tavakolpour S, Mahmoudi H, et al. Lichen planopilaris: retrospective study on the characteristics and treatment of 291 patients. J Dermatolog Treat. 2019; 30(6): 598–604. doi:10.1080/09546634.2018.1542480
- Chiang C, Sah D, Cho BK, et al. Hydroxychloroquine and lichen planopilaris: efficacy and introduction of Lichen Planopilaris Activity Index scoring system. J Am Acad Dermatol. 2010; 62(3): 387-392. doi:10.1016/j.jaad.2009.08.054
- Alirezaei P, Ahmadpanah M, Rezanejad A, et al. Compared to Controls, Individuals with Lichen Planopilaris Have More Depression, a Lower Self-Esteem, and a Lower Quality of Life. Neuropsychobiology. 2019; 78(2): 95-103. doi:10.1159/000499135
- Jha AK. Platelet-rich plasma as an adjunctive treatment in lichen planopilaris. J Am Acad Dermatol. 2019; 80(5): e109-e110. doi:10.1016/j.jaad.2018.09.013
- Plante J, Eason C, Snyder A, et al. Tofacitinib in the treatment of lichen planopilaris: a retrospective review [published online ahead of print, 2020 May 28]. J Am Acad Dermatol. 2020; S0190-9622(20)30979-8. doi:10.1016/j.jaad.2020.05.104
- Lajevardi V, Salarvand F, Ghiasi M, et al. The efficacy and safety of oral low dose naltrexone versus placebo in the patients with lichen planopilaris: a randomized controlled clinical trial [published online ahead of print, 2020 Jun 8]. J Dermatolog Treat. 2020; 1-5. doi:10.1080/09546634.2020.1774488
- Lee B, Elston DM. The uses of naltrexone in dermatologic conditions. J Am Acad Dermatol. 2019; 80(6): 1746-1752. doi:10.1016/j.jaad.2018.12.031
- Bolduc C, Sperling LC, Shapiro J. Primary cicatricial alopecia: Lymphocytic primary cicatricial alopecias, including chronic cutaneous lupus erythematosus, lichen planopilaris, frontal fibrosing alopecia, and Graham-Little syndrome. J Am Acad Dermatol. 2016; 75(6): 1081-1099.
- Vano-Galvan S, Trindade de Carvalho L, Saceda-Corralo D, et al. Oral minoxidil improves background hair thickness in lichen planopilaris [published online ahead of print, 2020 Apr 11]. J Am Acad Dermatol. 2020; S0190-9622(20)30566-1. doi:10.1016/j.jaad.2020.04.026
- Tegtmeyer K, Zhao J, Maloney NJ, et al. Off-label studies on tofacitinib in dermatology: a review [published online ahead of print, 2019 Oct 3]. J Dermatolog Treat. 2019; 1–11. doi:10.1080/09546634.2019.1673877
- Montilla AM, Gomez-Garcia F, Gomez-Arias PJ, et al. Scoping Review on the Use of Drugs Targeting JAK/STAT Pathway in Atopic Dermatitis, Vitiligo, and Alopecia Areata. Dermatol Ther (Heidelb). 2019; 9(4): 655–683. doi:10.1007/s13555-019-00329-y
- Hosking AM, Juhasz M, Mesinkovska NA. Topical Janus kinase inhibitors: A review of applications in dermatology. J Am Acad Dermatol. 2018; 79(3): 535–544. doi:10.1016/j.jaad.2018.04.018
- Shreberk-Hassidim R, Ramot Y, Zlotogorski A. Janus kinase inhibitors in dermatology: A systematic review. J Am Acad Dermatol. 2017; 76(4): 745-753.e19. doi:10.1016/j.jaad.2016.12.004
- Xeljanz/Xeljanz XR (tofacitinib) [package insert]. New York, NY: Pfizer Laboratories Division of Pfizer Inc. https://labeling.pfizer.com/showlabeling.aspx?id=959; 2019. Accessed July 7, 2020.
- FDA. Safety trial finds risk of blood clots in the lungs and death with higher dose of tofacitinib (Xeljanz, Xeljanz XR) in rheumatoid arthritis patients; FDA to investigate. https://www.fda.gov/drugs/drug-safety-and-availability/safety-trial-finds-risk-blood-clots-lungs-and-death-higher-dose-tofacitinib-xeljanz-xeljanz-xr; 2019. Accessed July 6, 2020.
