Nail Psoriasis and Psoriatic Arthritis for the Dermatologist
Rebecca Liu1, Braden M. Candela2, Joseph C English III2*
1University of Pittsburgh School of Medicine
2Department of Dermatology, University of Pittsburgh, Pittsburgh, PA
Abstract
Psoriatic arthritis (PsA) may affect up to a third of patients with psoriasis. It is characterized by diverse clinical phenotypes and as such, is often underdiagnosed, leading to disease progression and poor outcomes. Nail psoriasis (NP) has been identified as a risk factor for PsA, given the anatomical connection between the extensor tendon and nail matrix. Therefore, it is important for dermatologists to screen patients exhibiting symptoms of NP for joint manifestations. On physical exam, physicians should be evaluating for concurrent skin and nail involvement, enthesitis, dactylitis, and spondyloarthropathy. Imaging modalities, including radiographs and ultrasound, may also be helpful in diagnosis of both nail and joint pathology. Physicians should refer to Rheumatology when appropriate. Numerous systemic therapies are effective at addressing both NP and PsA including DMARDs, biologics, and small molecule inhibitors. These treatments ultimately can inhibit the progression of inflammatory disease and control symptoms, thereby improving quality of life for patients.
Introduction
Psoriasis is a chronic inflammatory disease that affects up to 3% of the population1. Of those patients, 30% may develop psoriatic arthritis (PsA), a complex disease characterized by diverse clinical phenotypes2-6. Among psoriasis patients followed by dermatology, about 15% were found to have undiagnosed PsA5. One study found that a delay of just 6 months in diagnosis can contribute to peripheral joint erosions and poor functional outcomes7.
The pathogenesis of PsA, similar to psoriasis, involves the complex dysregulation of T-cell subtypes (Th1, 2, 17, 19, Treg) and other dendritic cells producing pro-inflammatory cytokines that affect synovial fibroblasts, chondrocytes, and osteoblasts3. PsA is most common in North America, with a peak age of onset at >50 years of age and no differences in gender2. It can present peripherally, with variable joint distributions, and/or with axial involvement. Distinguishing features of PsA include enthesitis (inflammation at the site of tendon insertion into bone) and dactylitis (inflammation of two or more consecutive joints in the same finger). Enthesitis is observed in 30-50% of PsA patients, and is most commonly found at the Achilles tendon and plantar fascia. Some theories postulate that biomechanical stress at the entheses may result in the release of cytokines, leading to an articular inflammatory response and contributing to the pathogenesis of PsA8. Dactylitis occurs in 40-50% of patients, most commonly in the toes, and is associated with more severe disease8.
Risk factors for developing PsA include both genetic and clinical factors. Family history is important to obtain as first-degree relatives of patients with PsA have up to a 55 times higher risk of developing the disease2. Genetic markers have been identified on the human leucocyte antigen (HLA) regions with HLA-B*27 serving as a marker for disease severity. HLA-Cw6 is the most common susceptibility marker for psoriasis, but was found to be a negative predictor of PsA2,4. Other risk factors for PsA include obesity, metabolic abnormalities, smoking, alcohol use, as well as the early onset of extensive psoriatic skin disease especially scalp, gluteal cleft, and nail involvement2.
Nail psoriasis (NP)(Figure 1) has been found to be associated with an increased risk for PsA and can predict enthesopathy4,9. It has been reported to occur in up to 79% of patients with psoriasis but up to 86% of patients with PsA10,11. NP can involve multiple parts of the nail including the nail bed, nail matrix, or both. Nail bed disease may manifest as subungual hyperkeratosis, oil drop discoloration, splinter hemorrhages, pustules, and onycholysis. Nail matrix disease may present with nail plate crumbling, pitting, leukonychia, and red spots on the lunula. These symptoms can be associated with pain, psychological distress, and even interfere with activities of daily living, making it important to identify and treat appropriately12.
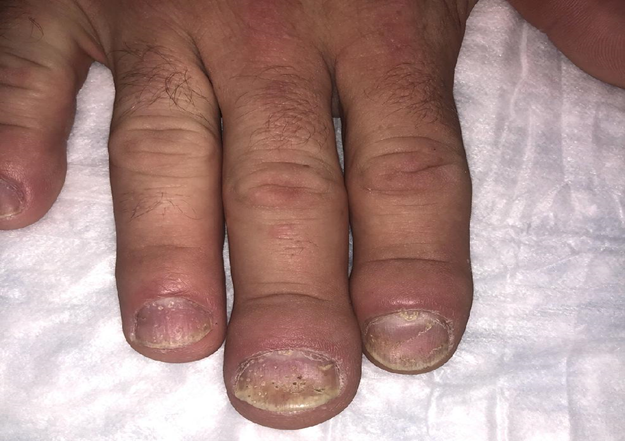
Figure 1: Nail psoriasis with early swelling of the distal tip of finger. Radiographic imaging demonstrated early distal PsA.
Diagnosis
With the diagnosis of NP, dermatologists should also examine for additional psoriatic skin involvement as well as concurrent PsA. When examining, assess for dactylitis by comparing the affected versus unaffected hands and feet, which helps distinguish between normal joint size and shape from abnormal joint swelling and sausage digit formation. Enthesitis can be examined by applying pressure to entheseal points (knee, elbow, pelvis, spine, ribcage, shoulder, Achilles tendon, and plantar fascia). Assessment for spinal disease includes testing the patient’s neck for range of motion in all directions. If restriction in movement is suspected, a validated assessment measure such as the modified Schober’s test or lateral lumbar flexion test is warranted13.
Several screening questionnaires have also been developed to assist in the early detection of PsA including the Toronto PsA Screen (ToPAS), Psoriasis Arthritis Screening Questionnaire (PASQ) and the Psoriasis Epidemiology Screening Tool (PEST). Formal classification criteria was proposed in 2006 based on the Classification of Psoriatic Arthritis (CASPAR) study which considers the presence of skin or nail involvement, dactylitis, negative rheumatoid factor, and juxtaarticular bone formation14.
Imaging modalities may be helpful to improve diagnostic accuracy. Radiographs can show joint erosions, joint space narrowing, bony proliferation, osteolysis (pencil in cup deformity), ankylosis, and spondylitis15. Recently, musculoskeletal ultrasound (US) has been studied as a non-invasive and inexpensive modality to assess for joint and tendon inflammation as well as nail disease13. Given the distal extensor tendon’s close association with the nail matrix, inflammation of the enthesis in the early phase of disease can affect the nail. This results in clinically significant pathology that can be identified with US16,17. Nail plate alternations can be seen with US in a majority of PsA patients, even in up to 75% of clinically normal appearing nails18. PsA patients have significantly increased nail matrix and nail bed thickness when compared to controls18,19. One study even recommended the use of US to examine the DIP joints in patients who had at least 5 dystrophic nails9. Further research is required for guidelines on how to use US in the clinical setting for diagnosis or monitoring of disease progression.
Dermatologists should feel comfortable doing a brief physical exam for patients who complain of joint pain and/or present with NP, obtaining imaging studies, and initiating a timely referral to Rheumatology. Understanding and identifying features of PsA at the time of presentation will help more patients get access to disease modifying treatments and prevent disabling disease progression.
Management
Management may depend on whether patients present with isolated or concomitant nail and joint involvement, as well as the severity of disease. Figure 2 identifies treatments for NP and PsA as well as the overlap of therapies with efficacy for both. This article will focus on exploring treatment options to address concurrent psoriatic nail and joint disease.
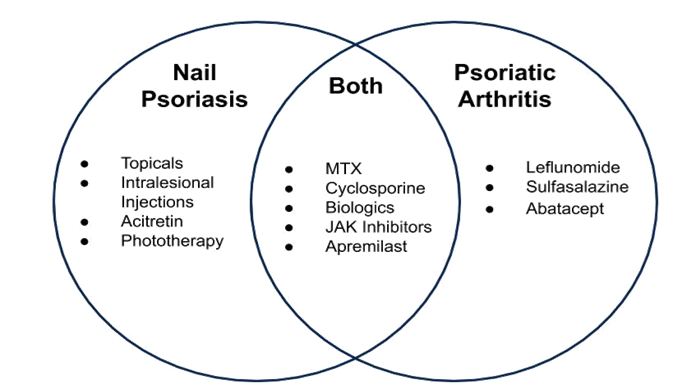
Figure 2: Treatment options for Nail Psoriasis and Psoriatic Arthritis
When discussing evidence based therapies for both NP and PsA, studies have used indexes such as the Nail Psoriasis Severity Index (NAPSI), and the American College of Rheumatology (ACR) criteria to evaluate response to treatment. For NAPSI, each nail is divided into four quadrants and evaluated for nail bed and nail matrix symptoms.
One nail can score up to 8 points, for a maximum of 80 among 10 fingernails20. ACR assesses for a certain percentage (20%/50%/70%) of improvement in tender and swollen joint counts, patient and physician global assessments, pain, disability, and acute phase reactant measures21.
Traditional DMARDs
Methotrexate (MTX) is one of the most common first line disease modifying anti-rheumatic drugs (DMARDs) used to treat PsA that also has efficacy for NP12,22. Cyclosporine, a calcineurin inhibitor, is typically reserved for more refractory PsA and limited to short-term use (up to 12 months), given its association with nephrotoxicity, hypertension, and malignancy12,23.
Biologic Therapies
2018 recommendations from the American College of Rheumatology (ACR) and National Psoriasis Foundation (NPF) suggest utilizing TNF alpha inhibitors (TNFi) even prior to DMARDs for treatment-naïve patients24. These TNFi include adalimumab, etanercept, infliximab, golimumab, and certolizumab pegol, all of which have shown great efficacy in treating both NP and PsA, including decreasing radiographic progression12,25. While no studies have compared the efficacy of all 5 TNFi on NP and PsA, there is data demonstrating nonsignificant differences between select TNFi26-28. Notably, combination therapy of biologics (etanercept, infliximab, golimumab) and MTX have shown superior efficacy compared to MTX monotherapy29-31. When selecting a TNFi, physicians should consider the patient’s preference for route of administration, frequency of treatment, and insurance coverage.
IL-17 and IL-23 inhibitors
Psoriasis research has identified the central roles of the IL-17 and IL-23 pathways for both NP and PsA pathogenesis, and thereby developed targeted anti-interleukin therapies. IL-17 inhibitors currently available include secukinumab and ixekizumab, both with significant efficacy in addressing NP and peripheral articular symptoms, in addition to inhibiting radiographic progression12,32. When compared to a TNFi (adalimumab), by 24 weeks ixekizumab showed a significantly greater improvement in treating both PsA and NP33. Brodalumab is another anti-IL17 drug currently under investigation for PsA. Phase 2 studies found significant superiority over placebo with higher rates of ACR20 and ACR50. It has also been shown to significantly improve NP by week 1212. Physicians should note that some patients receiving brodalumab reported suicidal ideation, although no causal relationship has been identified34,35. Therefore, we caution use in those with known psychiatric comorbidities. Ustekinumab is an IL-12 and IL-23 inhibitor that also demonstrated efficacy in treating NP and peripheral PsA with decreased radiographic progression12,36.
Other treatments that selectively target IL-23 have not been FDA approved for use in PsA; these include guselkumab, tildrakizumab, and risankizumab. Guselkumab has demonstrated efficacy for NP. Post hoc data analysis from VOYAGE 1 and VOYAGE 2 studies showed a significant improvement in NAPSI score over adalimumab by week 16. However, by 6 months, the magnitude of improvement did not differ between the two treatments37. A placebo-controlled phase 2 study with a primary endpoint of ACR20 for PsA showed significant improvement in symptoms as well38. Preliminary data from a phase 2B trial of tildrakizumab has shown significant efficacy in treating joint symptoms as early as 8 weeks, however limited data is available regarding its effect on NP39. Risankizumab, in a phase 2 head-to-head comparison with ustekinumab, had superior efficacy in measured secondary outcomes including NP and joint pain for PsA patients. Authors suggested that the blockade of the p19 subunit of IL-23, as opposed to the p40 subunit (blocked by ustekinumab), may have greater inhibited IL-23 activity, and improved efficacy of treatment40.
Phosphodiesterase Inhibitor
Apremilast is a phosphodiesterase 4 inhibitor that works intracellularly to downregulate many of the inflammatory mediators involved in psoriasis pathogenesis. NP and PsA trials have shown significant improvement in NAPSI and ACR20 scores by week 16. It is generally well tolerated and does not require laboratory monitoring12,41,42.
Small Molecule Inhibitors
Tofacitinib is an oral Janus kinase (JAK) inhibitor that can be used to treat both NP and PsA12,43. Its safety profile, however, is concerning for malignancy, risk of serious infection, and herpes zoster reactivation, which often limits its use clinically44,45. Other JAK inhibitors undergoing clinical trials for PsA include filgotinib and upadacitinib . Filgotinib, evaluated in EQUATOR, a multicenter phase 2 randomized trial, demonstrated efficacy in patients who had previously failed conventional DMARDs with improvements in psoriatic skin disease and enthesitis46. Upadacitinib is currently in phase 3 testing with no published data as of yet.
Treating Isolated NP
For NP without joint or significant cutaneous involvement, localized therapies may be considered to avoid the associated adverse effects of systemic medications. This includes corticosteroids (betamethasone), calcipotriol, retinoids (tazarotene and acitretin), calcineurin inhibitors (tacrolimus), as well as intralesional injections (triamcinolone acetone or MTX). Phototherapy options include pulse dye laser (PDL) psoralen–ultraviolet light A (PUVA), and narrowband ultraviolet B (NBUVB)12.
PsA Only Treatment
Some treatment options for PsA do not provide the additional benefit of addressing psoriatic nail disease. These include conventional DMARDs (leflunomide, sulfasalazine) and the immunomodulator abatacept, which we will not delve into further in this paper47.
Conclusion
Nail psoriasis should alert the dermatologist to further investigate for PsA. Examination for enthesitis, dactylitis, and spondyloarthropathy along with radiographic imaging and prompt referral to Rheumatology can improve the detection of PsA. Multiple new biologic therapies are available to treat both NP and PsA, decreasing the disease burden of afflicted patients.
Conflicts of Interest
The authors have no conflicts to declare.
References
- Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014; 70(3): 512-6.
- Solmaz D, Eder L, Aydin SZ. Update on the epidemiology, risk factors, and disease outcomes of psoriatic arthritis. Best Pract Res Clin Rheumatol. 2018; 32(2): 295-311.
- Veale DJ, Fearon U.The pathogenesis of psoriatic arthritis. Lancet. 2018; 391(10136): 2273-2284.
- Yan D, Ahn R, Leslie S, et al. Clinical and Genetic Risk Factors Associated with Psoriatic Arthritis among Patients with Psoriasis. Dermatol Ther (Heidelb). 2018; 8(4): 593-604.
- Villani AP, Rouzaud M, Sevrain M, et al. Prevalence of undiagnosed psoriatic arthritis among psoriasis patients: Systematic review and meta-analysis. J Am Acad Dermatol. 2015; 73(2): 242-8.
- Dominguez-Rosado I, Moutinho V Jr, DeMatteo RP, et al. Outcomes of the Memorial Sloan Kettering Cancer Center International General Surgical Oncology Fellowship. J Am Coll Surg. 2016; 222(5): 961-6.
- Haroon M1, Gallagher P1, FitzGerald O. Diagnostic delay of more than 6 months contributes to poor radiographic and functional outcome in psoriatic arthritis. Ann Rheum Dis. 2015; 74(6): 1045-50.
- Kehl AS, Corr M, Weisman MH. Review: Enthesitis: New Insights Into Pathogenesis, Diagnostic Modalities, and Treatment. Arthritis Rheumatol. 2016; 68(2): 312-22.
- Castellanos-González M, Joven BE, Sánchez J, et al. Nail involvement can predict enthesopathy in patients with psoriasis. J Dtsch Dermatol Ges. 2016; 14(11): 1102-1107.
- El Miedany Y, El Gaafary M, Youssef S, et al. Tailored approach to early psoriatic arthritis patients: clinical and ultrasonographic predictors for structural joint damage. Clin Rheumatol. 2015; 34(2): 307-13.
- Crowley JJ, Weinberg JM, Wu JJ, et al. Treatment of nail psoriasis: best practice recommendations from the Medical Board of the National Psoriasis Foundation. JAMA Dermatol. 2015; 151(1): 87-94.
- McClanahan DR1, English JC 3rd. Therapeutics for Adult Nail Psoriasis and Nail Lichen Planus: A Guide for Clinicians. Am J Clin Dermatol. 2018; 19(4): 559-584.
- Zhang A, Kurtzman DJB, Perez-Chada LM, et al. Psoriatic arthritis and the dermatologist: An approach to screening and clinical evaluation. Clin Dermatol. 2018. 36(4): 551-560.
- Mahmood F, Coates LC, Helliwell PS. Current concepts and unmet needs in psoriatic arthritis. Clin Rheumatol. 2018; 37(2): 297-305.
- Mease PJ, Armstrong AW. Managing patients with psoriatic disease: the diagnosis and pharmacologic treatment of psoriatic arthritis in patients with psoriasis. Drugs. 2014; 74(4): 423-41.
- Tan AL, Benjamin M, Toumi H, et al. The relationship between the extensor tendon enthesis and the nail in distal interphalangeal joint disease in psoriatic arthritis--a high-resolution MRI and histological study. Rheumatology (Oxford). 2007; 46(2): 253-6.
- Cunha JS, Qureshi AA, Reginato AM, Nail Enthesis Ultrasound in Psoriasis and Psoriatic Arthritis: A Report from the 2016 GRAPPA Annual Me.ting. J Rheumatol. 2017; 44(5): 688-690.
- Mondal S, Dutta S, Lahiri D, et al. Assessment of nail unit structures by ultrasound in patients with psoriatic arthritis and their correlations with disease activity indices: a case-control study. Rheumatol Int. 2018. 38(11): 2087-2093.
- Fassio A, Giovannini I, Idolazzi L, et al. Nail ultrasonography for psoriatic arthritis and psoriasis patients: a systematic literature review. Clin Rheumatol. 2019.
- Rich P, Scher RK. Nail Psoriasis Severity Index: a useful tool for evaluation of nail psoriasis. J Am Acad Dermatol. 2003; 49(2): 206-12.
- Felson DT, LaValley MP. The ACR20 and defining a threshold for response in rheumatic diseases: too much of a good thing. Arthritis Res Ther. 2014; 16(1): 101.
- Smolen JS, Landewé RBM, Bijlsma JWJ, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann Rheum Dis. 2017; 76(6): 960-977.
- Mahrle G, Schulze HJ, Färber L, et al. Low-dose short-term cyclosporine versus etretinate in psoriasis: improvement of skin, nail, and joint involvement. J Am Acad Dermatol. 1995; 32(1): 78-88.
- Singh JA, Guyatt G, Ogdie A, et al. Special Article: 2018 American College of Rheumatology/National Psoriasis Foundation Guideline for the Treatment of Psoriatic Arthritis. Arthritis Care Res (Hoboken). 2019; 71(1): 2-29.
- Goulabchand R, Mouterde G, Barnetche T, et al. Effect of tumour necrosis factor blockers on radiographic progression of psoriatic arthritis: a systematic review and meta-analysis of randomised controlled trials. Ann Rheum Dis. 2014; 73(2): 414-9.
- Saad AA, Symmons DP, Noyce PR, et al. Risks and benefits of tumor necrosis factor-alpha inhibitors in the management of psoriatic arthritis: systematic review and metaanalysis of randomized controlled trials. J Rheumatol. 2008; 35(5): 883-90.
- Ozmen I, Erbil AH, Koc E, et al. Treatment of nail psoriasis with tumor necrosis factor-alpha blocker agents: an open-label unblinded comparative study. J Dermatol; 2013; 40(9): 755-6.
- Sánchez-Regana M, Sola-Ortigosa J, Alsina-Gibert M, et al. Nail psoriasis: a retrospective study on the effectiveness of systemic treatments (classical and biological therapy). J Eur Acad Dermatol Venereol. 2011; 25(5): 579-86.
- Mease PJ, Gladman DD, Collier DH, et al. Etanercept and Methotrexate as Monotherapy or in Combination for Psoriatic Arthritis: Primary Results From a Randomized, Controlled Phase III Trial. Arthritis Rheumatol. 2019; 71(7): 1112-1124.
- Baranauskaite A, Raffayová H, Kungurov NV, et al. Infliximab plus methotrexate is superior to methotrexate alone in the treatment of psoriatic arthritis in methotrexate-naive patients: the RESPOND study. Ann Rheum Dis. 2012; 71(4): 541-8.
- Leonieke J J van Mens, Henriëtte M de Jong, Inka Fluri, et al., Achieving remission in psoriatic arthritis by early initiation of TNF inhibition: a double-blind, randomised, placebo-controlled trial of golimumab plus methotrexate versus placebo plus methotrexate. Ann Rheum Dis. 2019; 78(5): 610-616.
- Iain B McInnes, Philip J Mease, Bruce Kirkham, et al., Secukinumab, a human anti-interleukin-17A monoclonal antibody, in patients with psoriatic arthritis (FUTURE 2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2015; 386(9999): 1137-46.
- Mease PJ, Smolen JS, Behrens F, et al. A head-to-head comparison of the efficacy and safety of ixekizumab and adalimumab in biological-naive patients with active psoriatic arthritis: 24-week results of a randomised, open-label, blinded-assessor trial. Ann Rheum Dis. 2020; 79(1): 123-131.
- Blair HA. Brodalumab: A Review in Moderate to Severe Plaque Psoriasis. Drugs. 2018; 78(4): 495-504.
- Lebwohl MG, Papp KA, Marangell LB, et al. Psychiatric adverse events during treatment with brodalumab: Analysis of psoriasis clinical trials. J Am Acad Dermatol. 2018; 78(1): 81-89.e5.
- Rich P, Bourcier M, Sofen H, et al. Ustekinumab improves nail disease in patients with moderate-to-severe psoriasis: results from PHOENIX 1. Br J Dermatol. 2014; 170(2): 398-407.
- Foley P, Gordon K, Griffiths CEM, et al. Efficacy of Guselkumab Compared With Adalimumab and Placebo for Psoriasis in Specific Body Regions: A Secondary Analysis of 2 Randomized Clinical Trials. JAMA Dermatol. 2018; 154(6): 676-683.
- Deodhar A, Gottlieb AB, Boehncke WH, et al. Efficacy and safety of guselkumab in patients with active psoriatic arthritis: a randomised, double-blind, placebo-controlled, phase 2 study. Lancet. 2018; 391(10136): 2213-2224.
- Mease PJCS, Fructuoso FJG. Randomised, double-blind, placebo- controlled, multiple-dose, Phase 2B study to demonstrate the safety and efficacy of tildrakizumab, a high-affinity anti-interleukin-23p19 monoclonal antibody, in patients with active psoriatic arthritis. Abstract LB0002. 2019.
- Papp KA, Blauvelt A, Bukhalo M, et al. Risankizumab versus Ustekinumab for Moderate-to-Severe Plaque Psoriasis. N Engl J Med. 2017; 376(16): 1551-1560.
- Reich K, Gooderham M2, Green L, et al. The efficacy and safety of apremilast, etanercept and placebo in patients with moderate-to-severe plaque psoriasis: 52-week results from a phase IIIb, randomized, placebo-controlled trial (LIBERATE). J Eur Acad Dermatol Venereol. 2017; 31(3): 507-517.
- Wells AF, Edwards CJ, Kivitz AJ, et al. Apremilast monotherapy in DMARD-naive psoriatic arthritis patients: results of the randomized, placebo-controlled PALACE 4 trial. Rheumatology (Oxford). 2018; 57(7): 1253-1263.
- Merola JF, Elewski B, Tatulych S, et al. Efficacy of tofacitinib for the treatment of nail psoriasis: Two 52-week, randomized, controlled phase 3 studies in patients with moderate-to-severe plaque psoriasis. J Am Acad Dermatol. 2017; 77(1): 79-87.e1.
- Gladman D, Rigby W, Azevedo VF, et al. Tofacitinib for Psoriatic Arthritis in Patients with an Inadequate Response to TNF Inhibitors. N Engl J Med. 2017; 377(16): 1525-1536.
- Mease P, Hall S, FitzGerald O, et al., Tofacitinib or Adalimumab versus Placebo for Psoriatic Arthritis. N Engl J Med. 2017; 377(16): 1537-1550.
- Mease P, Coates LC, Helliwell PS, et al. Efficacy and safety of filgotinib, a selective Janus kinase 1 inhibitor, in patients with active psoriatic arthritis (EQUATOR): results from a randomised, placebo-controlled, phase 2 trial. Lancet. 2018; 392(10162): 2367-2377.
- Ritchlin CT, Colbert RA, Gladman DD. Psoriatic Arthritis. N Engl J Med. 2017; 376(10): 957-970.
