Commentary: Systemic Therapy for Mucosal Lichen Planus with a Focus on Oral Lichen Planus: Update and Review of Challenges and Successes
Roy S. Rogers, III, MD1; Suha Zawawi MBBS1*; Thais Pincelli MD1; Markéta Janovská MDDr.2; Alison Bruce MD1
1Department of Dermatology – Mayo Clinic Florida – Jacksonville, FL, USA
2Department of Oral Medicine, Institute of Dental Medicine, First Faculty of Medicine and General University Hospital, Charles University - Prague, Czech Republic.
Introduction
Lichen planus (LP) is a chronic inflammatory dermatosis of the skin, mucosa, hair, and nails. The sites of mucosal involvement include oral, genital, esophageal, ocular, otic, and less frequently bladder, nasal, laryngeal, and anal surfaces1. The skin and oral cavity are the two most common sites affected.
Oral lichen planus (OLP), a mucosal variant, tends to be chronic, sometimes involving multiple extra-oral sites, therapeutically challenging, and an oral potentially malignant disease (OPMD)1-4. The prevalence of OLP is estimated at 1-2%5. OLP is typically more chronic than cutaneous LP. Disease presenting primarily on the gingivae, known as desquamative gingivitis, affects approximately 10% of patients with OLP3,6.
No curative treatment for LP is available. Therapeutic approaches are directed at reducing inflammation and pain and improving the quality of life. As OLP is considered to be a T cell-mediated disease, therapeutic interventions have targeted the inflammatory pathways underlying OLP7.
Cutaneous LP may resolve spontaneously over several years, but spontaneous remission of mucosal LP is rare. Periodic follow-up of patients with OLP is indicated. If active disease persists, the patient should be monitored every 3-6 months. At the time of diagnosis of OLP, patients should be counseled about the increased risk of oral cancer and understand the need for periodic examinations even if they are asymptomatic, or if their symptoms are well-controlled2,4,8.
Mucosal Lichen Planus
The most common extra-oral location of mucosal lichen planus (MLP) is the genital mucosa1,3,9. Some patients have multiple mucosal sites involved simultaneously. Patients seldom associate oral symptoms of OLP with symptoms involving cutaneous, genital, or other mucosal sites and may feel uncomfortable when talking about these with their provider. Orogenital LP can affect both women and men1,10-12. Gingival involvement is a major component in these patients with the vulvovaginal-gingival and peno-gingival syndromes. As a consequence of chronic erosive and ulcerative mucosal LP scarring may cause major anatomic and functional problems including stenosis of the introitus and obstruction of the urethra. Patients may suffer intense pain with dysuria and dyspareunia1. Other mucosal surfaces such as esophageal, conjunctival, and otic tissues may be involved. Involvement in these areas causes great morbidity13-15.
The coexistence of OLP and vulvar lichen sclerosus (LS) is under-recognized. Both OLP and vulvar LS are common in older women. Janovska et al reported 86 women with both OLP and vulvar LS16. The preferred treatment was topical corticosteroids. Some patients with recalcitrant disease required systemic therapy. Drugs including hydroxychloroquine, doxycycline, metronidazole, methotrexate, and mycophenolate mofetil benefitted these patients. The use of systemic agents reduced the disease activity of both conditions and was thought to decrease the risk of developing oral and/or vulvar squamous cell carcinoma16.
Treatment of Mucosal Lichen Planus
Those LP patients with involvement of multiple mucosal sites are the most challenging to treat1,10-19. Multidisciplinary management with dermatologists, dentists and dental specialists, gastroenterologists, gynecologists, otorhinolaryngologists, and ophthalmologists involved in the ongoing care of patients with MLP may be necessary.
The main goal of therapy for MLP is the control of inflammation allowing healing and remission of the disease. Ancillary goals are control of symptoms of pain and the restoration of mucosal health. Treatment should be directed at eliminating atrophic, erosive, and ulcerative lesions and potentially lessening the likelihood of malignant transformation8,16,19.
Topical therapy is satisfactory for many patients with mild to moderate but limited mucosal disease7,9,17-20. Systemic therapy is indicated for those whose disease cannot be controlled with topical therapy, those whose disease is extensive and not amenable to topical therapy, and those whose disease involves multiple mucosal surfaces compromising organ function such as severe oral, esophageal, conjunctival, otic or genital involvement with scarring and chronic ulceration1,9,13-15,18,19.
Treatment of Mucosal Lichen Planus – Focus on Oral Lichen Planus
Therapy for OLP should begin with supportive measures such as gentle oral hygiene, control of gingivitis and oral candidiasis, smoking cessation, and limitation of alcohol intake, under the care of the patient’s dental practitioner. Topical therapy with fluorinated corticosteroids and calcineurin inhibitors, singly or in combination is the mainstay of treatment7,9,17-19.
The administration of systemic corticosteroids alone or as a bridge to other anti-inflammatory immunomodulating agents is an effective short-term treatment option. Systemic corticosteroids are used to control acute exacerbations and to achieve control of severe disease activity. Prednisone administered as a tapering 3-week course in a dose of 0.5 mg/kg in a single, daily dose for 1 week, tapered by 50% per week for the next two weeks is an effective starting point for systemic therapy of refractory MLP. Doses such as prednisone 40 mg/day for week 1, 20 mg/day for week 2, and 10 mg/day for week 3 would be appropriate for most patients7,18,19.
At the same time the systemic corticosteroid regimen is started, an anti-inflammatory immunomodulatory agent should be initiated to allow the agent time to exert its effect before corticosteroids are discontinued. Immunomodulatory therapies are numerous. There are no consensus treatment guidelines for systemic immunomodulatory therapy. We propose a treatment algorithm based on the review of existing evidence, recent literature, and the authors’ personal experience7,9,16-19,21. Fig.1
Figure 1:
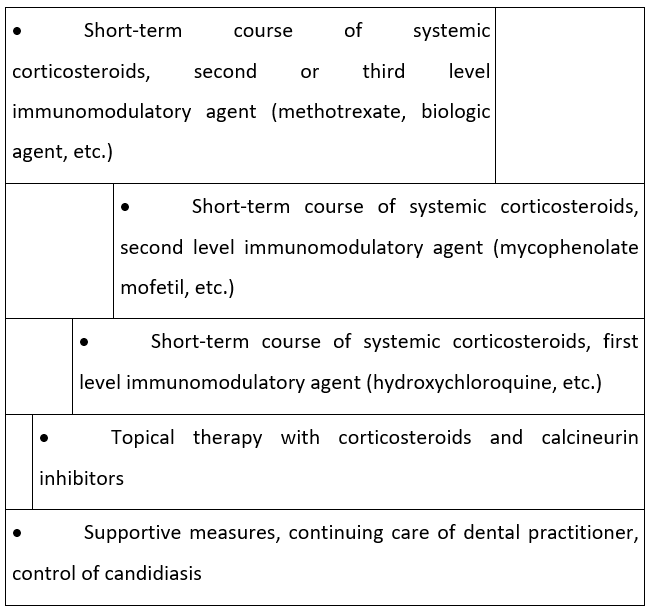
The first level of immunomodulatory agents are drugs that exert an anti-inflammatory effect in addition to their primary indication. Medications such as hydroxychloroquine, doxycycline, dapsone, and other antibiotics such as metronidazole and griseofulvin constitute these first-level systemic agents which should be started concomitantly to systemic corticosteroids.
Of these, hydroxychloroquine would be the first choice. Several trials with 10 patients22, 21 patients23, and a retrospective review24 support the use of hydroxychloroquine in MLP. A trial of 3-6 months should be undertaken. The clinician should expect that up to 65% of patients will achieve complete remission and the rest will have moderate to marked improvement with doses of 200-400 mg daily for 6 months. If improvement is not complete, a second first-level immunomodulatory agent such as doxycycline or metronidazole may be added for a 3-month trial16,25.
Treatment of Patients with Recalcitrant Mucosal Lichen Planus
Clinicians should expect that many patients with MLP who have been refractory to excellent oral hygiene, ongoing dental management, and supportive care, plus a tapering course of systemic corticosteroids and a 3–6-month trial of a first-level systemic immunomodulatory agent, will be challenging to treat. These are the most intractable cases requiring patience and dedication.
If the MLP is not controlled with first-level immunomodulatory agents, the clinician should seek improvement with second-level drugs19. Antimetabolites such as methotrexate (MTX), mycophenolate mofetil (MMF), or azathioprine are second-level options21,25-30. In this group, MMF would be our first choice. A 3–6-month trial with MMF in doses of 1000-3000 mg per day is appropriate.
The third level of immunomodulatory therapy includes biological agents such as rituximab, adalimumab, and etanercept19. Experience with these agents is limited to case reports and small case series. Despite the reports of certain biologic agents causing drug-induced lichenoid tissue reactions31-33, we consider biologics a new and emerging therapeutic option for MLP that will likely replace the older immunomodulatory drugs. The same way that some biologics are useful in treating inflammatory bowel disease while others may trigger it, a similar effect may be observed with biologics and MLP. These contradictory responses may reflect a balance of different immunity pathways. Recently, the therapy with an oral JAK-inhibitor, tofacitinib, resulted in complete remission in a patient with intractable MLP with severe esophageal involvement34.
Summary and Conclusions
MLP is a chronic debilitating, painful, and distressing disease. At this time, no curative treatment regimen exists. The current literature is replete with case reports, small series, and non-randomized trials of therapy. The goal of treatment is to provide comfort for the patient. Therapy should focus on controlling inflammation, halting the development of new lesions, and providing the opportunity for established lesions to heal. In the face of this chronic and scarring disease, effective therapy is mandatory. Our treatment algorithm begins with supportive measures under the care of the dental practitioner. This must be maintained throughout the time it may take to gain complete remission.
We recommend a treatment algorithm of escalating intensity of management beginning with a 3-week course of systemic corticosteroids at which time the first level immunomodulatory agent is initiated. The corticosteroid in a pulse fashion reduces the intensity of inflammation and permits the immunomodulator to start controlling disease activity.
Gaining and sustaining remission is challenging. However, the clinician should expect marked or moderate improvement if not complete remission in 3-6 months in many patients. Maintaining remission for 3 additional months before tapering the immunomodulatory agent is essential. Long-term, low-dose immunosuppressive therapy may be necessary.
Clinicians should be aware that flares while undergoing immunosuppressive therapy may represent an episode of acute candidiasis rather than an exacerbation of the MLP.
Refractory MLP may require second and third immunomodulatory agents for 3–6-month trials. We have suggested our preference for each level based on our review of the current literature and our personal experience in managing MLP patients.
As the old drugs (corticosteroids, hydroxychloroquine, MTX, and MMF) get gradually replaced by the newer and more targeted ones (JAK-inhibitors and biologics), the future of therapeutics in MLP becomes more promising. We believe that oral JAK-inhibitors such as tofacitinib and the new emerging biological agents will increase our therapeutic arsenal to manage this debilitating condition.
Conflicts of Interest: The authors have no conflicts of interest and no financial interest to report.
References
- Rogers RS, 3rd, Eisen D. Erosive oral lichen planus with genital lesions: the vulvovaginal-gingival syndrome and the peno-gingival syndrome. Dermatol Clin. Jan 2003; 21(1): 91-8, vi-vii. doi: 10.1016/s0733-8635(02)00059-1.
- Warnakulasuriya S, Kujan O, Aguirre-Urizar JM, et al. Oral potentially malignant disorders: A consensus report from an international seminar on nomenclature and classification, convened by the WHO Collaborating Centre for Oral Cancer. Oral Dis. Nov 2021; 27(8): 1862-1880. doi: 10.1111/odi.13704.
- Eisen D. The evaluation of cutaneous, genital, scalp, nail, esophageal, and ocular involvement in patients with oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. Oct 1999; 88(4): 431-6. doi: 10.1016/s1079-2104(99)70057-0.
- Eisen D. The clinical features, malignant potential, and systemic associations of oral lichen planus: a study of 723 patients. J Am Acad Dermatol. Feb 2002; 46(2): 207-14. doi: 10.1067/mjd.2002.120452.
- González-Moles M, Warnakulasuriya S, González-Ruiz I, et al. Worldwide prevalence of oral lichen planus: A systematic review and meta-analysis. Oral Dis. May 2021; 27(4): 813-828. doi: 10.1111/odi.13323.
- Rogers RS, 3rd, Sheridan PJ, Nightingale SH. Desquamative gingivitis: clinical, histopathologic, immunopathologic, and therapeutic observations. J Am Acad Dermatol. Dec 1982; 7(6): 729-35. doi: 10.1016/s0190-9622(82)70153-7.
- Lodi G, Manfredi M, Mercadante V, et al. Interventions for treating oral lichen planus: corticosteroid therapies. Cochrane Database Syst Rev. Feb 28, 2020; 2(2): Cd001168. doi: 10.1002/14651858.CD001168.pub3.
- Laniosz V, Torgerson RR, Ramos-Rodriguez AJ, et al. Incidence of squamous cell carcinoma in oral lichen planus: a 25-year population-based study. International journal of dermatology. Mar 2019; 58(3): 296-301. doi: 10.1111/ijd.14215.
- Eisen D. The clinical manifestations and treatment of oral lichen planus. Dermatol Clin. Jan 2003;21(1):79-89. doi:10.1016/s0733-8635(02)00067-0.
- Pelisse M, Leibowitch M, Sedel D, et al. [A new vulvovaginogingival syndrome. Plurimucous erosive lichen planus]. Ann Dermatol Venereol. 1982; 109(9): 797-8. Un nouveau syndrome vulvo-vagino-gingival. Lichen plan érosif plurimuqueux.
- Pelisse M. The vulvo-vaginal-gingival syndrome. A new form of erosive lichen planus. International journal of dermatology. Jul-Aug 1989; 28(6): 381-4. doi: 10.1111/j.1365-4362.1989.tb02484.x
- Cribier B, Ndiaye I, Grosshans E. [Peno-gingival syndrome. A male equivalent of vulvo-vagino-gingival syndrome?]. Rev Stomatol Chir Maxillofac. 1993; 94(3): 148-51. Syndrome péno-gingival. Un équivalent masculin du syndrome vulvo-vagino-gingival?
- Katzka DA, Smyrk TC, Bruce AJ, et al. Variations in presentations of esophageal involvement in lichen planus. Clin Gastroenterol Hepatol. Sep 2010; 8(9): 777-82. doi: 10.1016/j.cgh.2010.04.024.
- Sartori-Valinotti JC, Bruce AJ, Krotova Khan Y, et al. A 10-year review of otic lichen planus: the Mayo Clinic experience. JAMA Dermatol. Sep 2013; 149(9): 1082-6. doi: 10.1001/jamadermatol.2013.4711.
- Brewer JD, Ekdawi NS, Torgerson RR, et al. Lichen planus and cicatricial conjunctivitis: disease course and response to therapy of 11 patients. J Eur Acad Dermatol Venereol. Jan 2011; 25(1): 100-4. doi: 10.1111/j.1468-3083.2010.03693.x.
- Janovska M, Bodiford KJ, Bruce AJ, et al. Coexistence of oral lichen planus and vulvar lichen sclerosus. Oral Surg Oral Med Oral Pathol Oral Radiol. Mar 9 2023; doi: 10.1016/j.oooo.2023.02.017.
- Al-Hashimi I, Schifter M, Lockhart PB, et al. Oral lichen planus and oral lichenoid lesions: diagnostic and therapeutic considerations. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. Mar 2007; 103 Suppl: S25.e1-12. doi: 10.1016/j.tripleo.2006.11.001.
- Olson MA, Rogers RS, 3rd, Bruce AJ. Oral lichen planus. Clin Dermatol. Jul-Aug 2016; 34(4): 495-504. doi: 10.1016/j.clindermatol.2016.02.023.
- Pincelli T, Tolaymat L, Sluzevich J, et al. Systemic Medical and Procedural Therapies for Mucosal Lichen Planus: Update and Review of Challenges and Successes. Journal of Skin and Stem Cell. 2020; 7(4).
- Eisen D, Carrozzo M, Bagan Sebastian JV, et al. Number V Oral lichen planus: clinical features and management. Oral Dis. Nov 2005; 11(6): 338-49. doi: 10.1111/j.1601-0825.2005.01142.x.
- Marnach ML, Torgerson RR. Therapeutic Interventions for Challenging Cases of Vulvar Lichen Sclerosus and Lichen Planus. Obstet Gynecol. Sep 1, 2021; 138(3): 374-378. doi: 10.1097/aog.0000000000004498.
- Eisen D. Hydroxychloroquine sulfate (Plaquenil) improves oral lichen planus: An open trial. J Am Acad Dermatol. Apr 1993; 28(4): 609-12. doi: 10.1016/0190-9622(93)70082-5.
- Yeshurun A, Bergman R, Bathish N, et al. Hydroxychloroquine sulphate therapy of erosive oral lichen planus. Australas J Dermatol. May 2019; 60(2): e109-e112. doi: 10.1111/ajd.12948.
- Vermeer HAB, Rashid H, Esajas MD, et al. The use of hydroxychloroquine as a systemic treatment in erosive lichen planus of the vulva and vagina. Br J Dermatol. Jul 2021; 185(1): 201-203. doi:10.1111/bjd.19870.
- Husein-ElAhmed H, Gieler U, Steinhoff M. Lichen planus: a comprehensive evidence-based analysis of medical treatment. J Eur Acad Dermatol Venereol. Oct 2019; 33(10): 1847-1862. doi: 10.1111/jdv.15771.
- Lajevardi V, Ghodsi SZ, Hallaji Z, et al. Treatment of erosive oral lichen planus with methotrexate. J Dtsch Dermatol Ges. Mar 2016; 14(3): 286-93. doi: 10.1111/ddg.12636.
- Dunaway S, Tyler K, Kaffenberger J. Update on treatments for erosive vulvovaginal lichen planus. International journal of dermatology. Mar 2020; 59(3): 297-302. doi: 10.1111/ijd.14692.
- Verma KK, Mittal R, Manchanda Y. Azathioprine for the treatment of severe erosive oral and generalized lichen planus. Acta Derm Venereol. Oct-Nov 2001; 81(5): 378-9. doi: 10.1080/000155501317140197.
- Ashack KA, Haley LL, Luther CA, et al. Assessing the clinical effectiveness of an algorithmic approach for mucosal lichen planus (MLP): A retrospective review. J Am Acad Dermatol. Jun 2016; 74(6): 1073-1076.e2. doi: 10.1016/j.jaad.2015.11.024.
- Hrin ML, Bowers NL, Feldman SR, et al. Mycophenolate mofetil versus methotrexate for vulvar lichen planus: A 10-year retrospective cohort study demonstrates comparable efficacy and tolerability. J Am Acad Dermatol. Aug 2022; 87(2): 436-438. doi: 10.1016/j.jaad.2021.08.061.
- Giudice A, Liborio F, Averta F, et al. Oral Lichenoid Reaction: An Uncommon Side Effect of Rituximab. Case Rep Dent. 2019; 2019: 3154856. doi: 10.1155/2019/3154856.
- Andrade P, Lopes S, Albuquerque A, et al. Oral Lichen Planus in IBD Patients: A Paradoxical Adverse Effect of Anti-TNF-α Therapy. Dig Dis Sci. Sep 2015; 60(9): 2746-9. doi: 10.1007/s10620-015-3680-2.
- Asarch A, Gottlieb AB, Lee J, et al. Lichen planus-like eruptions: an emerging side effect of tumor necrosis factor-alpha antagonists. J Am Acad Dermatol. Jul 2009; 61(1): 104-11. doi: 10.1016/j.jaad.2008.09.032.
- Kozlov M, Levit EK, Silvers DN, et al. Severe Esophageal Lichen Planus Treated with Tofacitinib. 2023.
