Electronic Tattoos: A Promising Approach to Real-time Theragnostics
Nicholas X. Williams1, Aaron D. Franklin1,2*
1Department of Electrical and Computer Engineering, Duke University, Durham NC 27708, USA
2Department of Chemistry, Duke University, Durham NC 27708, USA
Abstract
Real-time monitoring of relevant biological signals, in combination with the timely delivery of target drugs, would be ideal for treating most medical conditions. However, access to biological fluids without a bulky, costly, and cumbersome apparatus remains challenging, as does the ability to deliver drugs of controlled dosage in a similarly unobtrusive fashion. The skin provides a promising medium for access and dosing using biomedical electronics, colloquially dubbed electronic tattoos. Recent developments in biologically compatible, flexible materials and devices have brought electronic tattoos closer to reality for sensing biomarkers extracted from the skin and delivering target drugs through the dermis. In this review, the materials and engineering requirements, fabrication developments, and sensing and therapeutic advancements of electronic tattoos are presented. Three components are required for a complete theragnostic electronic tattoo system: 1) supporting electronics for control and data transmission; 2) diagnostic sensors, categorized as mechanical (measure an internal stimulus) and chemical (measure a chemical change); and 3) therapeutics for drug delivery. The leading approaches for fabrication are summarized, including the transfer of flexible devices to the skin and the direct printing of devices onto the epidermis. Altogether, while significant obstacles remain, the advancements in this field show great promise for realizing electronic tattoo theragnostics to revolutionize point-of-care medicine.
Introduction and Background
One of the greatest shortcomings in modern medicine is the infrequent measurement of key biomarkers, particularly those within the blood, combined with the regimented dosing of drugs. Continuous monitoring of relevant analytes in tandem with the precise dosing of medication would have profound implications on treatment and health. From early diagnosis to improved long-term prognosis, low delay between the onset of symptoms and detection of an aberrant signal has the potential to revolutionize medicine1,2. This is readily apparent for chronic diseases, such as diabetes, where it has long been understood that frequent collection of data on current blood sugar levels is pivotal to proper management of symptoms3. Countless other chronic and acute diseases, such as Crohn’s disease and complications due to heart failure, could be transformed with similar capabilities; yet, the closest we have come to widespread continuous health monitoring is the recent proliferation of wearables, such as smartwatches, which allow for facile, non-invasive monitoring of limited ex vivo patient data, such as heart rate, daily movement, and sleep4,5. Thanks to advancements in biocompatible materials and sensors, the scope of devices for continuous monitoring and chronic symptom regulation is expanding rapidly. As numerous biological signals (including both chemical and physical signals) become more accessible to measurement, the capability for targeted and timely drug delivery upon detection of a specific biological trigger progresses closer to reality.
There are numerous modalities that support continuous monitoring and dosing, including wearables (such as a smart watch or smart clothing), ingestibles or implantables, and on-skin electronics (also referred to as electronic tattoos, epidermal electronics, and e-tattoos). While wearables are currently the most ubiquitous option, their utility is somewhat limited by their form factor. To maintain comfort during use, minimal direct contact with the skin is made, thus detection of chemical (e.g., biomarkers in blood) and physical (e.g., strain) biological signals may be limited6,7. Ingestible electronic biomonitoring technology is unsurpassed in its ability to detect chemical signals within the native biological environment, but is intrinsically transient and difficult to control location post ingestion8,9. Further, any in vivo technology, including implantable electronics, will be wrought with challenges of biofouling due to an immunogenic foreign body response, which can lead to discomfort, signal drift, and the rejection of the device10.
Electronic tattoos are nonpermanent electrical devices or systems placed in intimate contact with the skin and intended for relatively short-term use (upwards of 1-2 weeks). Their name is derived from their similarity to temporary, decal-style tattoos rather than an ink embedded into the dermis to change local pigmentation. While there may be some minimally invasive penetration into the skin, the vast majority of the device, including all electronics, are on top of the skin. Electronic tattoos combine the comfort and less transient nature of wearables with much of the precision of ingestibles; an on-the-skin technology with the promise of advancing the current diagnosis-only model to one that combines therapy and diagnostics for a complete “theragnostic” system. Electronic tattoos have intimate contact with the skin, and thus the ability to directly monitor biological signals through the epidermal layer in addition to transferring a therapeutic drug via the dermis. Their relative thinness would allow these tattoo-like biomedical devices to be comfortable to wear, with semi-permanence in that they have been demonstrated to be stable in performance for over a week of continuous usage11. Their lifetime is frequently considered to be a few days at the shortest12 to the cycle length of desquamation of the outermost layer of the dermis, which is about 20-30 days13.
To facilitate the development of electronic tattoos for continuous monitoring and therapy, three separate components must work in concert with one another: biological sensing / diagnostics; drug delivery / therapeutics; and a support system to facilitate the functioning of all components (Figure 1). While sensing and dosing frequently retain focal eminence, the support system (including electrodes, processing, encapsulation, and filtering) is no less pivotal to the functionality. Herein, we describe the recent progress across these three areas in the field of electronic tattoos for theragnostics.
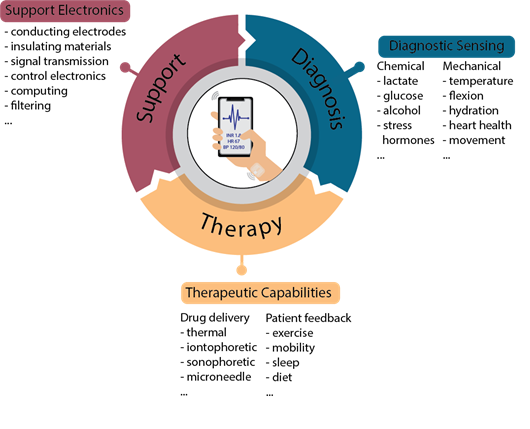
Figure 1: Schematic of modules required for on-skin, electronic tattoos for theragnostics. For each module, an example list of biomarkers (diagnostics), approaches (therapeutics), or components (supporting system) is provided. Some degree of unified functionality across these modules is critical to realizing biomedical electronic tattoos.
Support Electronics
Without a system to support and facilitate transduction and communication, the implementation of on-skin sensing and therapy would be all but impossible. Numerous considerations must be made to ensure accuracy, comfort, and durability. Human skin can stretch by up to 25% before incurring damage14. Given this, to maintain comfort, an epidermal tattoo must be thin enough and have a sufficiently low modulus (< 1.5 mm thick and < 600 kilopascals (kPa)) to retain comfort and must maintain performance metrics when strained15.
One major concern is loss of electrical conductivity due to strain; to alleviate this failure mechanism, numerous methods have been proposed, starting with material selection. Recently, significant work has been performed on liquid metals for stretchable conductive electronics16,17. At room temperature, metals such as eutectic indium-gallium maintain a liquid form, which allows conductive channels containing these materials to be stressed over 300% without any noticeable degradation18. This, however, has the drawback of requiring encapsulation, which greatly reduces utility and versatility. Another materials category, that perhaps is more directly suited to electronic tattoos, are high aspect ratio materials, such as silver nanowires. Their high aspect ratio allows for only a small increase in electrical resistance with applied bending and tensile strain19,20, and makes them an ideal candidate for electrodes and contacts in an electronic tattoo21.
In addition to materials choice, significant improvements to lifetime and resistance to degradation from cyclic strain can be achieved via engineering design of electrodes through the incorporation of a serpentine path. This approach can increase ultimate strain from as little as 1% for some materials to upwards of 300%22. This design alleviates stress from tensile strain as it allows for the coiled electrodes to straighten before considerable stress is placed upon the conductive trace itself. To further resist degradation, encapsulation can also work to sustain performance by upwards of 6x after numerous cyclic strain episodes typical of quotidian movement23.
Finally, in addition to conductive traces that are the backbone of electronics, signal processing and transmission are required. Most conventional electronic medical devices separate on- or in-body measurement from amplification, signal filtering, processing, and interpretation; however, in some cases for fully on-skin devices, these processes must be miniaturized and incorporated into the electronic tattoo package. While the vast majority of demonstrated electronic tattoos use conventional silicon-based integrated circuits (ICs)19,24, there is a growing body of research developing stretchable transistors and other stretchable electrical components for incorporation into flexible electronics25,26. Currently, the performance of conventional electronics is orders of magnitude greater and the scale is substantially smaller than achievable with flexible components. Thus the incorporation of flexible circuit boards may require an increase in footprint as compared to a silicon IC27. However, if conformity to the skin is a requirement, a device fabricated from all flexible components may be desired, in which case, the larger area could be less of a concern.
The most likely path for the support electronics will be to first implement only the needed control circuitry and signal transmission to an external device (e.g., smartphone, as depicted in Figure 1), keeping the complexity on-skin to a minimum; then, in the longer term, implement other support electronics into the tattoo when the feasibility of doing so is realized. Even in the near-term, obstacles remain for realizing all needed diagnostic and therapeutic control along with signal transmission in an on-skin electronic tattoo, including in the performance and stability of the electronic devices, electrical interconnections and interfaces, and scalability in cost and size.
Diagnostic Sensing
There are numerous cases where continuous monitoring can be advantageous as compared to discrete point monitoring, not least of which is the removal of required human interfacing, which can be hindered by exhaustion, misuse of tools, and/or access to a clinic. With new technological advancements propelling both consumer electronics and medical devices towards an internet-of-everything (IOE) ideal of connectivity, research into electronic biomedical sensors has proliferated. Much of the development focus is in on-skin sensors. From its birth in 1999 with the continuous detection of blood glucose28, the field has expanded to include sensing of strain29,30 (Figure 2A-B), temperature31 (Figure 2C) and several other biological markers related to the exponential increase in sweat sensors32,33 in the past several years. These on-skin electrical sensors can be divided into two equally necessary categories: physical and chemical sensors.
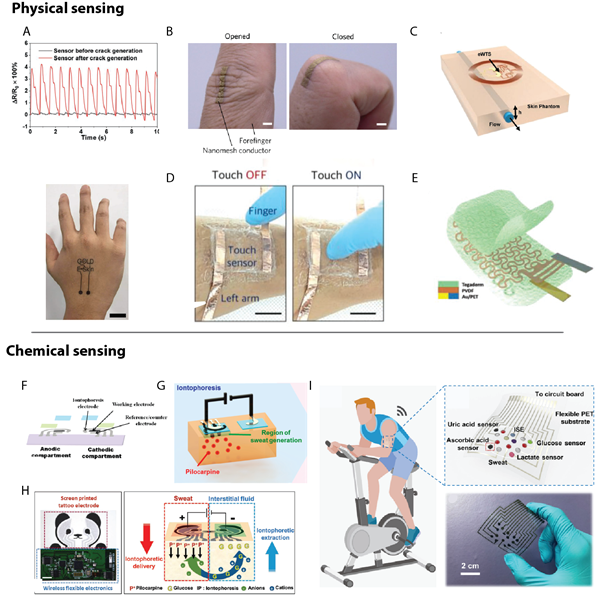
Figure 2: Types of electronic tattoo diagnostic sensors. Physical sensors, which use a physical change in the sensor to measure a biological signal, including (A, B) strain sensors and (C) thermal sensors, (D) touch sensors, and (E) electrocardiograms. Chemical sensors measure the response of chemical reactions such as (F) blood glucose and (G) alcohol. Multiplexed sensors to measure both (H) alcohol & blood glucose and (I) sweat analytes. Reprinted with permission from: (A) ref. 29, copyright 2019, Advanced Materials; B) ref. 30, copyright 2017, Nature Nanotechnology; C) ref. 31, copyright 2019, Advanced Science; D) ref. 12, copyright 2018, Advanced Functional Materials; E) ref. 22, copyright 2019, Advanced Science; F) ref. 34, copyright 2015, Analytical Chemistry; G) ref. 35, copyright 2016, ACS sensors; H) ref. 36, copyright 2018, Advanced Science; I) ref 37, copyright 2019, Science Advances).
Physical and chemical sensors can be distinguished by the sensing mechanism. Physical sensors measure an attribute via a change to the sensor itself, whereas chemical sensors measure a reaction between the target analyte and the sensor. As an example, physical sensors include temperature sensors and strain sensors. For a temperature sensor, the sensing mechanism is derived from a change in resistance of the active sensing component in response to a modification in skin temperature. A strain sensor, likewise, transduces the strain experienced by the skin (through compression, extension, or torsion) via a proportional change in the resistance caused by some physical change to the sensor. Depending on the sensor itself, this could be a capacitive change as in a touch sensor12 (Figure 2D), an increase or decrease to the junction density between electrically conductive components (such as silver nanowires)30, or modulation to energy band gap in the channel region of a transistor34, among others. The commonality between these transduction mechanisms is an internal change to a structure or property of the sensor itself. Hence, an electrocardiogram (ECG), would likewise be categorized as a physical sensor as the electrical signal is generally measured via a capacitive change22 (Figure 2E).
Chemical sensors, on the other hand, transduce information (most often, electrically) via the response to a chemical shift. This may be with an oxidoreductase enzymatic reaction (such as glucose oxidation facilitated by glucose oxidase, as seen in Figure 2F-H)35-37 or via the binding event of a protein-protein pair (such as the immunogenic antibody-antigen binding of an immunoassay)38-40. While physical sensors can measure signals transdermally, chemical sensors require a solute, and hence a bodily fluid. This can be achieved ex vivo via sweat41 (Figure 2I) or interstitial fluid42,43.
For interstitial fluid – the fluid between cells within the body – cultivation requires microneedles to puncture the epidermis, while sweat sensing can be achieved without any poration of the skin. As with blood, interstitial fluid is easily probed with an implantable device; however, it is difficult to access non-invasively44, whereas sweat can be easily generated and measured transdermally. There are two methods for generation of sweat: natural production33 and the more common induction via the iontophoretic delivery of a drug, such as Pilocarpine45. While natural methods may be ideal for monitoring sweat during exercise, stimulated perspiration allows for greater control over sweat timing and volume. Sweat generation remains an issue for long-term use as natural techniques cannot be maintained due to physical exertion requirements and induced perspiration uses a limited drug resource on a localized device. Furthermore, due to degradation of the sensing mechanism, much of the testing focuses on rapid detection on the order of seconds29,35,36,41,46,47, minutes24,37,48,49, and hours50, rather than stability over days of use. To propel chemical sensing development into a commercializable stratum, more focus is needed on extending the lifetime of sensors from hours to days.
Given the relative novelty of this field, many of the reports are proof-of-concept and thus require further testing to directly compare to commercial sensors. To overcome this barrier, significant efforts are currently underway to increase the sensitivity, selectivity, and reproducibility of chemical biosensors51. Simultaneous delivery of all three of these metrics is required and will reduce anguish caused by false positives and delayed treatment caused by false negatives52. One significant hurdle, as of yet not entirely overcome, is lack of reproducibility in electronic biosensors at least partially caused by drift during storage or intrinsic device-to-device variations53. Furthermore, as the field advances, validation studies comparing electronic tattoos to the gold standards of clinic-based detection to ensure the correct and accurate measurement of analytes will become imperative. The field is new enough that this morass has not yet affected development because much of the work to date focuses on initial, singular demonstrations54; however, more detailed and long-term studies should become a more expected element of future studies. Recent efforts have seen success in on-chip calibration, which compares the response from a blank to that of a functionalized device on a single chip55, yet more research and validation are required to extend this development to more devices.
In contrast to chemical sensors, much of the limitations of current physical sensors are largely related to degradation due to repetitive sensing and to cyclic bending/stretching56-58. As previously stated, human skin can be strained significantly before damage14. Recent studies indicate that a 90° wrist flexion can strain the epidermal surface of the forearm by upwards of 25%59,60. Given that these are minor movements that occur continually throughout daily motion, cyclic strain reliability is a major issue. While numerous reports include some cyclic strain data, the majority fall short of truly substantive findings, given that most perform tests to below 1000 cycles58,61-66 or perform cyclic tests to low strain rates64. As the field progresses, cycle number in cyclic degradation testing must also increase if these epidermal electronics are to be used for the entire lifecycle of the epidermis. While there are still significant impediments to overcome, non-invasive, continuous monitoring has the potential to bring a momentous leap in disease control when combined with a feedback loop-based therapy system.
Therapeutic Capabilities
In conjunction with a diagnostic sensing and support system, transdermal drug delivery via an epidermal tattoo has the potential for non-invasive therapy as well as patient-specific regulation of drug delivery67. The skin is constituted by three layers: the waterproof epidermis, which is made up of keratinocytes, melanocytes, merkel and Langerhans cells; the middle layer or dermis, consisting of hair follicles, sweat and sebaceous glands, nerves, collagen, lymph vessels, and blood vessels; and the last layer, the hypodermis, which consists of the subcutaneous fat layer. The outermost layer of the epidermis, the stratum corneum (SC), acts as an efficacious barrier membrane, limiting diffusion of large molecules to the dermis67.
Multiple methods, both physical and chemical, have been developed to increase diffusion of a target drug through the SC, the most prominent of which are thermal enhancement68 (Figure 3A), which uses localized vasodilation from directly applied heat that can increase blood flow to a specified area by upwards of 9x, causing a 13x increase in uptake of a drug, such as nicotine69. The elevated heat can lead to some discomfort for the user and it is also difficult to predict delivery rates, which could lead to the delivery of potentially fatal drug concentrations70. A more controlled method is iontophoretic delivery (Figure 3B)71,72, which uses an applied voltage to transport charged drugs across the SC via electrophoresis and electroosmosis, allowing for upwards of a 10x improvement in drug diffusion over thermal delivery68; however, this method does require large, possibly dangerous voltages to achieve desirable delivery rates. Another method is microneedle delivery (Figure 3D)73,74, which uses small needles to penetrate through the SC; this poration allows for a desired drug to bypass the SC and migrate directly to the dermis. Microneedle is frequently used with another method to further enhance performance. In addition, another technique is sonophoresis75-77 (Figure 3E), which uses ultrasound to either heat the skin or increase permittivity via acoustic cavitation and generally requires a large ultrasonic transducer incompatible with electronic tattoos, and finally chemically enhanced diffusion (Figure 3F)78, which increases permittivity of the desired drug through encapsulation and thus is difficult to accurately dose in timed increments79. Regardless of the delivery mechanism, transdermal drug delivery requires a sensing component to take the requisite readings, (as seen in Figure 3G). Of all these options for skin-based drug delivery that can be electronically controlled, iontophoresis shows the greatest promise for electronic tattoos.
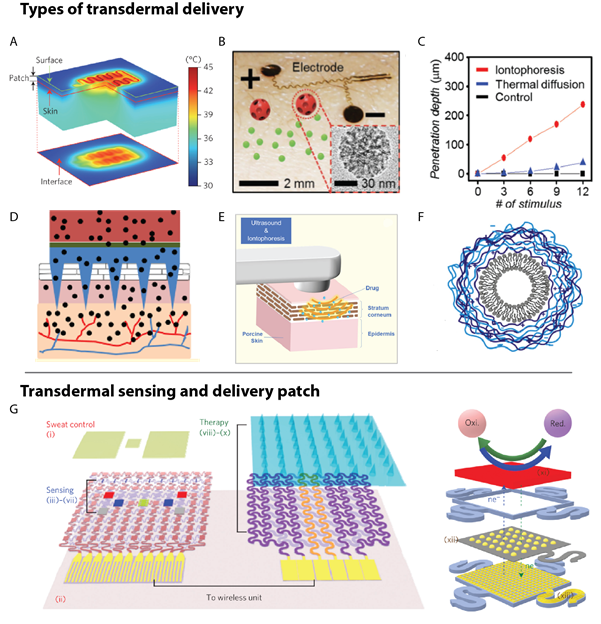
Figure 3: Electronic tattoo-based transdermal drug delivery. Delivery methods for diffusion of drugs through the SC layer of the epidermis include (A) thermal (using heat to increase diffusion), (B-C) iontophoresis (using an electro-repulsive force), (D) microneedle (puncturing the SC layer), (E) sonophoresis (using ultrasonic enhancement), and (F) chemical/ encapsulation (coating the desired drug to increase diffusion). (G) A schematic of a complete patch demonstrates the incorporation of a diagnostic sensing component and a therapeutic component that contains one or more of the delivery enhancement strategies. Reprinted with permission from: (A) ref. 80, copyright 2014, Nature Nanotechnology; B-C) ref. 68, copyright 2016, Advanced Healthcare Materials; D) ref. 73, copyright 2013, Molecular Pharmaceutics; E) ref. 77, copyright 2018, Advanced Drug Delivery Reviews; F) ref. 79, copyright 2015, Colloids Surfaces B: Biointerfaces; G) ref. 46, copyright 2016, Nature Nanotechnology).
Iontophoretic drug delivery offers a promising method for incorporation into an electronic tattoo system due to its electronic operation, especially when combined with poration of the epidermis using microneedles80,81, and elimination of the discomfort associated with elevated temperatures incident to thermal diffusion enhancement82. Iontophoresis enhances drug diffusion through the skin with an induced electric field. This method functions via electrophoresis and electroosmosis, which allows for transportation of larger molecules (>500 Da) previously blocked by the SC83. The strength of the electric field directly controls diffusion, and thus iontophoretic delivery can fully control dosage and dosing intervals. However, many embodiments of iontophoretic delivery require non-ideal conditions to enhance drug dosing. Given that the resistivity of human skin is between 1,000 to 100,000 Ω84, frequently high voltages are required for even modest currents required for diffusion enhancement. While numerous publications solely report currents, the publications that do report voltages use staggeringly high voltages of between 30-90V. These voltages are of such a magnitude to lyse red blood cells84,85 and would be difficult to implement in standalone electronics; hence, further research is required to decrease the electric field strength required to increase diffusion. One possible route to accomplish this is by decreasing the electrode gap, thus decreasing the electrical resistance of the system. While the remaining challenges are significant, motivation for complete electronic theragnostic epidermal tattoo systems is high and thus warrants further research into solutions for an on-skin therapeutic drug delivery system.
Fabrication and Scalability
One main differentiator distinguishing epidermal electronics from conventional wearables is the application method. While wearables are generally incorporated into a rigid electrical device, such as a wristwatch, epidermal electronics necessitate intimate contact to the skin, and thus must be flexible and stretchable. These limitations enforce constraints on materials and design selection, both of which limit processing options. There are two subsets of fabrication: transfer method, where the tattoo is fabricated onto a disposable substrate and transferred to the desired location on the skin in a similar manner to placing a decal-style tattoo or a sticker37,86,87; and direct printing, wherein the device is printed directly onto the epidermis21,88. For a directly printed device, this can be achieved either via a 3D printing technique, such as an extrusion method like FDM, or via a tradition printing technique using an inkjet or aerosol jet printer to print the temporary tattoo directly onto the skin. The transfer method offers a large array of fabrication approaches that include conventional cleanroom techniques, which allow for high performing devices through well-established processing technologies29,41,47,89 (Figure 4A-B); however, these processing technologies are costly and compromise the needed scalability in cost for bespoke electronic biomedical tattoos. Whereas printed electronics30,90-92 (Figure 4C-D) allow for the custom fabrication of low-cost components93 with challenges related more to the performance of the printed devices.
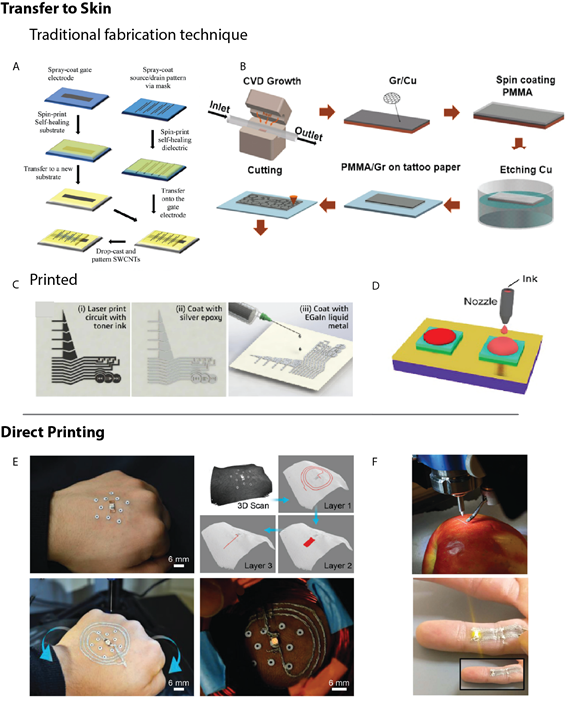
Figure 4: Fabrication techniques for electronic tattoos. The transfer method involves fabrication onto a disposable substrate either via (A-B) traditional, cleanroom fabrication technique schematic process flow or (C-D) via a printing method where the material is deposited in a solution form onto the transfer substrate. (E-F) The direct printing technique involves deposition of the electrically active inks directly onto the desired biological tissue. Reprinted with permission from: (A) ref. 90, copyright 2018, Small; B) ref. 89, copyright 2017, ACS Nano; C) ref. 92, copyright 2018, ACS Applied Materials & Interfaces; D) ref. 91, copyright 2018, Nature; E) ref. 88, copyright 2018, Advanced Materials; F) ref. 21, copyright 2019, Nanoscale).
Direct printing allows for customization to the patient’s needs and rapid prototyping because there is no delay between fabrication and utilization. Direct printing also eliminates the potential for errors in transferring, which plague alternate methods. Yet, direct printing is not yet a drop-in replacement for traditional fabrication methods and their transfer to skin, as deposition of electronically active materials and inks directly onto biological tissue of nearly all printed electronics requires caustic or otherwise damaging post-processing to achieve the desired electrical properties94. Recently, research interest has grown in the area of printable electrically conductive inks that can be cured at low temperatures95, with a subset that allow for desired performance at biologically compatible temperatures96. Yet, even so, the conductivity of these is often orders of magnitude below that of their bulk equivalents21. The growing interest in in-place-printed electronics also provides promising developments for the incorporation of more complex components into direct printed epidermal tattoos21,88 (Figure 4E-F).
Further development is still required to eliminate biologically incompatible temperatures and toxic chemicals and a substantial amount of progress is needed for other electronic materials and, ultimately, devices to be printed onto skin. Hence, direct printed electronic tattoos are currently limited to only a few uses that focus on electrically conductive inks for the fabrication of both sensing and supportive components21,88. Even so, both demonstrations have used silver nanomaterials, which are known to be cytotoxic and, as with other metal nanoparticles, may alter immune responses97 and might thus eventually cause cancer98,99. While silver nanowires are less cytotoxic than silver nanoparticles, frequent use still remains a concern, especially with the incorporation of heating and microneedles to increase penetration into the dermis as the reduction of elemental silver (Ag0) to ionic silver (Ag+) is at least a contributing factor to its toxicity and heating may increase the reaction rate while poration of the skin will increase exposure to the dermis100. As research progresses, a greater focus must be placed on the development of biocompatible inks, both in terms of ink constituents and long-term effects of the resultant printed films. Direct printing could be a powerful tool for expanding the utility of electronic tattoos beyond the one-size-fits-all model that prevails with transferred on-skin electronics. It’s likely that the first implementation of biomedical electronic tattoos would include some combination of transferred and directly printed components.
Even with completely biocompatible materials, further complications such as allergic contact dermatitis (ACD) may arise from frequent use of a patch in intimate contact with the skin. Along with increased discomfort from skin irritation, frequent exposure to a specific allergen could lead to skin sensitization, which is an immunological response101. The T cell mediated inflammatory response could potentially skew the results, or cause complete failure, of the intended sensing capabilities that prompted the use of the electronic tattoo in the first place due to biofouling, as occurs with implantable glucose sensors for diabetes patients102. Thus, considerable research must be performed on large populations to assess the viability and general safety of the extended use of electronic tattoos for theragnostic purposes.
Conclusion and Outlook
Significant work has recently been performed to develop the three systems required for transdermal theragnostics – supporting electronics, diagnostic systems, and therapeutic delivery. Advancements in support electronics allow for comfortable use through stretchable electronic materials and engineering design developments; yet, more work is required to enhance the performance of stretchable support electronics to a level commensurate with the rigid silicon-based devices currently on the market. Diagnostic systems, especially the monitoring of analytes in sweat, have garnered significant interest recently, and monumental advancements in detection breadth and sensitivity have been achieved in both chemical and physical sensing. However, improvements in reproducibility and degradation resistance still require focused development to improve accuracy and duration, two metrics that must be improved in future transdermal sensing devices. Finally, therapeutic delivery, when used in tandem with diagnostic systems and facilitated by support electronics, can seamlessly maintain homeostasis through targeted and automated transdermal, iontophoretic drug delivery; however, to realize these advancements, the power requirements of iontophoresis must be reduced to increase the safety of these devices.
In order to fabricate these on-skin devices, two separate methods can be used: the devices can be fabricated on a disposable substrate and transferred to the skin or the devices can be printed directly onto the skin. Transfer deposition allows for a broader range of fabrication methods, including traditional, cleanroom fabrication techniques; this could allow for high-performance devices given the materials choices available. However, transferring has the potential to introduce errors and cleanroom fabrication is costly, particularly for custom electronics. Direct printing alleviates this potential for error and offers a scalable, low-cost approach, but currently is limited in scope due to the small number of inks that are compatible with low-temperature deposition. In order to move towards direct printing, significant focus must be spent on the development of a wide array of inks compatible with direct deposition onto the skin.
Electronic tattoos have the potential to shift the paradigm of medical testing towards continuous monitoring, allowing for more rapid, potentially lifesaving, treatment in addition to a simplified cycle of care that eliminates much of the complexities involved with discrete point monitoring systems. This type of real-time monitoring is required for the incorporation of fast-acting and customized therapeutic systems that can, for instance, deliver a customized dose of target drugs in response to biological signals. While more developments are necessary, electronic tattoos have the potential to revolutionize the point-of-care landscape by incorporating a continuous and non-invasive feedback loop into theragnostics, decreasing the time to care and transforming long-term prognosis.
References
- Becker DP, Miller JD, Ward JD, et al. The outcome from severe head injury with early diagnosis and intensive management. J Neurosurg. 1977; 47: 491–502.
- Mueller C, Twerenbold R, Reichlin T. Early diagnosis of myocardial infarction with sensitive cardiac troponin assays. Clin Chem. 2019; 65: 490–491.
- European Diabetes Policy Group. A desktop guide to Type 2 diabetes mellitus. European Diabetes Policy Group 1999. Diabet Med. 1999; 16: 716–730.
- Alharbi M, Bauman A, Neubeck L, et al. Validation of Fitbit-Flex as a measure of free-living physical activity in a community-based phase III cardiac rehabilitation population. Eur J Prev Cardiol. 2016; 23: 1476–1485.
- Moreno-Pino F, Porras-Segovia A, López-Esteban P, et al. Validation of Fitbit Charge 2 and Fitbit Alta HR Against Polysomnography for Assessing Sleep in Adults With Obstructive Sleep Apnea. J Clin Sleep Med. 2019; 15: 1645–1653.
- Wong CK, Mentis HM, Kuber R. The bit doesn’t fit: Evaluation of a commercial activity-tracker at slower walking speeds. Gait Posture. 2018; 59: 177–181.
- O'Connell S, ÓLaighin G, Quinlan LR. When a step is not a step! Specificity analysis of five physical activity monitors. PLoS One. 2017; 12: 1–14.
- van der Schaar PJ, Dijksman JF, Broekhuizen-de Gast H, et al. A novel ingestible electronic drug delivery and monitoring device. Gastrointest Endosc. 2013; 78: 520–528.
- Bettinger CJ. Materials Advances for Next-Generation Ingestible Electronic Medical Devices. Trends Biotechnol. 2015; 33: 575–585.
- Wiley Encyclopedia of Biomedical Engineering. John Wiley & Sons Inc 2006. doi:10.1002/9780471740360
- Madhvapathy SR. Epidermal Electronic Systems for Measuring the Thermal Properties of Human Skin at Depths of up to Several Millimeters. Adv Funct Mater. 2018; 28: 1–11.
- Seo JW, Kim H, Kim K, et al. Calcium-Modified Silk as a Biocompatible and Strong Adhesive for Epidermal Electronics. Adv Funct Mater. 2018; 1800802: 1–13.
- Wilhelm KP, Cua AB, Maibach HI. Skin Aging: Effect on Transepidermal Water Loss, Stratum Corneum Hydration, Skin Surface pH, and Casual Sebum Content. Arch Dermatol. 1991; 127: 1806–1809.
- Widlund T, Yang S, Hsu YY, et al. Stretchability and compliance of freestanding serpentine-shaped ribbons. Int J Solids Struct. 2014; 51: 4026–4037.
- Jeong JW, Kim MK, Cheng H, et al. Capacitive epidermal electronics for electrically safe, long-term electrophysiological measurements. Adv Healthc Mater. 2014; 3: 642–648.
- Andrews JB, Mondal K, Neumann TV, et al. Patterned Liquid Metal Contacts for Printed Carbon Nanotube Transistors. ACS Nano. 2018; 12: 5482–5488.
- Votzke C, Daalkhaijav U, Menguc Y, et al. 3D-Printed Liquid Metal Interconnects for Stretchable Electronics. IEEE Sens J. 2019; 19: 3832–3840.
- Dickey MD. Stretchable and Soft Electronics using Liquid Metals. Adv Mater. 2017; 29: 1–19.
- Chun S. Conductive and Stretchable Adhesive Electronics with Miniaturized Octopus-Like Suckers against Dry/Wet Skin for Biosignal Monitoring. Adv Funct Mater. 2018; 1805224: 1805224.
- Liang J, Tong K, Pei Q. A Water-Based Silver-Nanowire Screen-Print Ink for the Fabrication of Stretchable Conductors and Wearable Thin-Film Transistors. Adv Mater. 2016; 28: 5986–5996.
- Williams NX , Noyce S , Cardenas JA , et al. Silver nanowire inks for direct-write electronic tattoo applications. Nanoscale. 2019; 11: 14294–14302.
- Ha T, Tran J, Liu S, et al. A Chest-Laminated Ultrathin and Stretchable E-Tattoo for the Measurement of Electrocardiogram, Seismocardiogram, and Cardiac Time Intervals. Adv Sci. 2019; 6.
- Li K. A Generic Soft Encapsulation Strategy for Stretchable Electronics. Adv Funct Mater. 2019; 29: 1–12.
- Mishra RK. Wearable potentiometric tattoo biosensor for on-body detection of G-type nerve agents simulants. Sensors Actuators B Chem. 2018; 273: 966–972.
- Wang Q. Self-Healable Multifunctional Electronic Tattoos Based on Silk and Graphene. Adv Funct Mater. 2019; 1808695: 1808695.
- Andrews JB. Fully Printed and Flexible Carbon Nanotube Transistors for Pressure Sensing in Automobile Tires. IEEE Sens J. 2018; 18: 7875–7880.
- Myny K. The development of flexible integrated circuits based on thin-film transistors. Nat Electron. 2018; 1: 30–39.
- Montagnana M, Lippi G, Guidi GC. Continuous glucose monitoring and type 1 diabetes. N Engl J Med. 2009; 360: 190–192.
- Gong S, Yap LW, Zhu B, et al. Local Crack-Programmed Gold Nanowire Electronic Skin Tattoos for In-Plane Multisensor Integration. Adv Mater. 2019; 1903789: 1903789.
- Miyamoto A, Lee S, Cooray NF, et al. Inflammation-free, gas-permeable, lightweight, stretchable on-skin electronics with nanomeshes. Nat Nanotechnol. 2017; 12: 907–913.
- Krishnan SR, Su CJ, Xie Z, et al. Wireless , Battery-Free Epidermal Electronics for Continuous , Quantitative , Multimodal Thermal Characterization of Skin. Small. 2018; 1803192: 1–13.
- Tai LC, Gao W, Chao M, et al. Methylxanthine Drug Monitoring with Wearable Sweat Sensors. Adv Mater. 2018; 30: 1–8.
- Parlak O, Keene ST, Marais A, et al. Molecularly selective nanoporous membrane-based wearable organic electrochemical device for noninvasive cortisol sensing. Sci Adv. 2018; 4.
- Kanaparthi S, Badhulika S. Solvent-free fabrication of a biodegradable all-carbon paper based field effect transistor for human motion detection through strain sensing. Green Chem. 2016; 18: 3640–3646.
- Bandodkar AJ, Jia W, Yardimci C, et al. Tattoo-based noninvasive glucose monitoring: A proof-of- concept study. Anal Chem. 2015; 87: 394–398.
- Kim J. Noninvasive Alcohol Monitoring Using a Wearable Tattoo-Based Iontophoretic-Biosensing System. ACS Sensors. 2016; 1: 1011–1019.
- Kim J, Sempionatto JR, Imani S, et al. Simultaneous Monitoring of Sweat and Interstitial Fluid Using a Single Wearable Biosensor Platform. Adv Sci. 2018; 5.
- White SP, Frisbie CD, Dorfman KD. Detection and Sourcing of Gluten in Grain with Multiple Floating-Gate Transistor Biosensors. ACS Sensors. 2018; 3: 395–402.
- Williams NX, Watson N, Joh DY, et al. Aerosol jet printing of biological inks by ultrasonic delivery. Biofabrication. 2020; 12: 025004.
- Joh DY, Hucknall AM, Wei Q, et al. Inkjet-printed point-of-care immunoassay on a nanoscale polymer brush enables subpicomolar detection of analytes in blood. Proc Natl Acad Sci. 2017; 7054–7062. doi:10.1073/pnas.1703200114
- He W, Wang C, Wang H, et al. Integrated textile sensor patch for real-time and multiplex sweat analysis. Sci Adv. 2019; 5: 1–9.
- Windmiller JR, Zhou N, Chuang MC, et al. Microneedle array-based carbon paste amperometric sensors and biosensors. Analyst. 2011; 136: 1846–1851.
- Matzeu G, Florea L, Diamond D. Advances in wearable chemical sensor design for monitoring biological fluids. Sensors Actuators B Chem. 2015; 211: 403–418.
- Bariya M, Nyein HYY, Javey A. Wearable sweat sensors. Nat Electron. 2018; 1: 160–171.
- Emaminejad S, Gao W, Wu E, et al. Autonomous sweat extraction and analysis applied to cystic fibrosis and glucose monitoring using a fully integrated wearable platform. Proc Natl Acad Sci U S A. 2017; 114: 4625–4630.
- Lee H, Choi TK, Lee YB, et al. A graphene-based electrochemical device with thermoresponsive microneedles for diabetes monitoring and therapy. Nat Nanotechnol. 2016; 11: 566–572.
- Zhang S. Hydrogel-Enabled Transfer-Printing of Conducting Polymer Films for Soft Organic Bioelectronics. Adv Funct Mater. 2019; 1906016: 1906016.
- Parrilla M, Guinovart T, Ferré J, et al. A Wearable Paper-Based Sweat Sensor for Human Perspiration Monitoring. Adv Healthc Mater. 2019; 8: 1–6.
- Rose DP, Ratterman ME, Griffin DK, et al. Adhesive RFID sensor patch for monitoring of sweat electrolytes. IEEE Trans Biomed Eng. 2015; 62: 1457–1465.
- Choi DH, Kim JS, Cutting GR, et al. Wearable potentiometric chloride sweat sensor: The critical role of the salt bridge. Anal Chem. 2016; 88: 12241–12247.
- Gao Z. Graphene transistor arrays functionalized with genetically engineered antibody fragments for Lyme disease diagnosis. 2D Mater. 2020; 7: 024001.
- Skaik Y. Understanding and using sensitivity, specificity and predictive values. Indian J Ophthalmol. 2008; 56: 341.
- Zhang X, Jing Q, Ao S, et al. Ultrasensitive Field-Effect Biosensors Enabled by the Unique Electronic Properties of Graphene. Small. 2019; 1902820: 1902820.
- Piccinini E. Pushing the Boundaries of Interfacial Sensitivity in Graphene FET Sensors: Polyelectrolyte Multilayers Strongly Increase the Debye Screening Length. J Phys Chem C. 2018; 122: 10181–10188.
- Gutiérrez-Sanz Ó. Transistor-based immunosensing in human serum samples without on-site calibration. Sensors Actuators B Chem. 2019; 295: 153–158.
- Jensen DW, Pascual J. Degradation of graphite/bismaleimide laminates with multiple embedded fiber optic sensors. Fiber Opt Smart Struct Ski III. 1990; 1370: 228.
- Yamada T, Hayamizu Y, Yamamoto Y, et al. A stretchable carbon nanotube strain sensor for human-motion detection. Nat Nanotechnol. 2011; 6: 296–301.
- Amjadi M, Pichitpajongkit A, Lee S, et al. Highly stretchable and sensitive strain sensor based on silver nanowire-elastomer nanocomposite. ACS Nano. 2014; 8: 5154–5163.
- Maiti R, Gerhardt LC, Lee ZS, et al. In vivo measurement of skin surface strain and sub-surface layer deformation induced by natural tissue stretching. J Mech Behav Biomed Mater. 2016; 62: 556–569.
- Muñoz MJ, Bea JA, Rodríguez JF, et al. An experimental study of the mouse skin behaviour: Damage and inelastic aspects. J Biomech. 2008; 41: 93–99.
- Valentine AD, Busbee TA, Boley JW, et al. Hybrid 3D Printing of Soft Electronics. Adv Mater. 2017; 29: 1–8.
- Rahimi R, Ochoa M, Yu W, et al. Highly stretchable and sensitive unidirectional strain sensor via laser carbonization. ACS Appl Mater Interfaces. 2015; 7: 4463–4470.
- Frutiger A, Muth JT, Vogt DM, et al. Capacitive soft strain sensors via multicore-shell fiber printing. Adv Mater. 2015; 27: 2440–2446.
- Kim SR, Kim JH, Park JW. Wearable and Transparent Capacitive Strain Sensor with High Sensitivity Based on Patterned Ag Nanowire Networks. ACS Appl Mater Interfaces. 2017; 9: 26407–26416.
- Xu H , Lv Y , Qiu D , et al. An ultra-stretchable, highly sensitive and biocompatible capacitive strain sensor from an ionic nanocomposite for on-skin monitoring. Nanoscale. 2019; 11: 1570–1578.
- Zhong J. Stretchable Self-Powered Fiber-Based Strain Sensor. Adv Funct Mater. 2015; 25: 1798–1803.
- Anselmo AC, Gokarn Y, Mitragotri S. Non-invasive delivery strategies for biologics. Nat Rev Drug Discov. 2018; 18: 19–40.
- Choi MK, Park OK, Choi C, et al. Cephalopod-Inspired Miniaturized Suction Cups for Smart Medical Skin. Adv Healthc Mater. 2016; 5: 80–87.
- Petersen KK, Rousing ML, Jensen C, et al. Effect of local controlled heat on transdermal delivery of nicotine. Int J Physiol Pathophysiol Pharmacol. 2011; 3: 236–242.
- Hao J, Ghosh P, Li SK, et al. Heat effects on drug delivery across human skin. Expert Opin Drug Deliv. 2016; 13: 755–768.
- O'Neil EC, Huang J, Suhler EB, et al. Iontophoretic delivery of dexamethasone phosphate for non-infectious, non-necrotising anterior scleritis, dose-finding clinical trial. Br J Ophthalmol. 2018; 102: 1011–1013.
- Byrne JD, Jajja MR, Schorzman AN, et al. Iontophoretic device delivery for the localized treatment of pancreatic ductal adenocarcinoma. Proc Natl Acad Sci U S A. 2016; 113: 2200–2205.
- Kochhar JS, Lim WX, Zou S, et al. Microneedle integrated transdermal patch for fast onset and sustained delivery of lidocaine. Mol Pharm. 2013; 10: 4272–4280.
- Cheung K, Das DB. Microneedles for drug delivery: trends and progress. Drug Deliv. 2016; 23: 2338–2354.
- Park D, Won J, Shin UC, et al. Transdermal drug delivery using a specialized cavitation seed for ultrasound. IEEE Trans Ultrason Ferroelectr Freq Control. 2019; 66: 1057–1064.
- Seah BCQ, Teo BM. Recent advances in ultrasound-based transdermal drug delivery. Int J Nanomedicine. 2018; 13: 7749–7763.
- Park J, Lee H, Lim GS, et al. Enhanced Transdermal Drug Delivery by Sonophoresis and Simultaneous Application of Sonophoresis and Iontophoresis. AAPS PharmSciTech. 2019; 20: 1–7.
- Herman A, Herman AP. Essential oils and their constituents as skin penetration enhancer for transdermal drug delivery: A review. J Pharm Pharmacol. 2015; 6:7 473–485.
- Jeon S, Yoo CY, Park SN. Improved stability and skin permeability of sodium hyaluronate-chitosan multilayered liposomes by Layer-by-Layer electrostatic deposition for quercetin delivery. Colloids Surfaces B Biointerfaces. 2015; 129: 7–14.
- Lee H. Device-assisted transdermal drug delivery. Adv Drug Deliv Rev. 2018; 127: 35–45.
- Alkilani AZ, McCrudden MT, Donnelly RF. Transdermal drug delivery: Innovative pharmaceutical developments based on disruption of the barrier properties of the stratum corneum. Pharmaceutics. 2015; 7: 438–470.
- Son D, Lee J, Qiao S, et al. Multifunctional wearable devices for diagnosis and therapy of movement disorders. Nat Nanotechnol. 2014; 9: 397–404.
- Bos JD, Meinardi MM. The 500 Dalton rule for the skin penetration of chemical compounds and drugs. Exp Dermatol. 2000; 9: 165–169.
- Wei Z, Zheng S, Wang R, et al. A flexible microneedle array as low-voltage electroporation electrodes for in vivo DNA and siRNA delivery. Lab Chip. 2014; 14: 4093–4102.
- Choi SO, Kim YC, Park JH, et al. An electrically active microneedle array for electroporation. Biomed. Microdevices. 2010; 12: 263–273.
- Wang Y. Low-cost, μm-thick, tape-free electronic tattoo sensors with minimized motion and sweat artifacts. npj Flex Electron. 2018; 2: 6.
- Brossard FSF, Pecunia V, Ramsay AJ, et al. Inkjet Printed Nanocavities on a Photonic Crystal Template. 2015; 17253: 17253.
- Zhu Z, Guo SZ, Hirdler T, et al. 3D Printed Functional and Biological Materials on Moving Freeform Surfaces. Adv Mater. 2018; 1707495: 1707495.
- Kabiri Ameri S, Ho R, Jang H, et al. Graphene Electronic Tattoo Sensors. ACS Nano. 2017; 11: 7634–7641.
- Khatib M, Huynh TP, Deng Y, et al. A Freestanding Stretchable and Multifunctional Transistor with Intrinsic Self-Healing Properties of all Device Components. Small. 2018; 1803939: 1803939.
- Wang S, Xu J, Wang W, et al. Skin electronics from scalable fabrication of an intrinsically stretchable transistor array. Nature. 2018; 555: 83–88.
- Lopes PA, Paisana H, De Almeida AT, et al. Hydroprinted Electronics: Ultrathin Stretchable Ag-In- Ga E-Skin for Bioelectronics and Human-Machine Interaction. ACS Appl Mater Interfaces. 2018; 10: 38760–38768.
- Subramanian V. Printed Electronics For Low-Cost Electronic Systems : Technology Status and Application Development. 2008; 17–24.
- Lee YK, Kim J, Kim Y, et al. Room Temperature Electrochemical Sintering of Zn Microparticles and Its Use in Printable Conducting Inks for Bioresorbable Electronics. Adv Mater. 2017; 1702665: 1702665.
- Han YD. The fabrication of highly conductive and fl exible Ag patterning through baking Ag nanosphere − nanoplate hybrid ink at a low temperature of 100 ° C. Nanotechnology. 2018; 29: 135301.
- Yang X, Sun M, Bian Y, et al. A Room-Temperature High-Conductivity Metal Printing Paradigm with Visible-Light Projection Lithography. Adv Funct Mater. 2018; 1807615: 1807615.
- Schrand AM. Metal-based nanoparticles and their toxicity assessment. Wiley Interdiscip Rev Nanomedicine Nanobiotechnology. 2010; 2: 544–568.
- Grivennikov SI, Greten FR, Karin M. Immunity inflammation and cancer. Cell. 2010; 140: 883–899.
- Kim MJ, Shin S. Toxic effects of silver nanoparticles and nanowires on erythrocyte rheology. Food Chem Toxicol. 2014; 67: 80–86.
- Volkov Y. Autophagy induction by silver nanowires: A new aspect in the biocompatibility assessment of nanocomposite thin films. Toxicol Appl Pharmacol. 2012; 264: 451–461.
- Nassau S, Fonacier L. Allergic Contact Dermatitis. Med Clin North Am. 2020; 104: 61–76.
- Klueh U1, Kaur M, Montrose DC, et al. Infammation and glucose sensors: Use of dexamethasone to extend glucose sensor function and life span in vivo. J Diabetes Sci Technol. 2007; 1: 496–504.
